Table of contents
In approximately 30% of cases, epilepsy cannot be adequately treated with pharmaceutical drugs, which is why essential oils are gaining attention as a complementary treatment.
What needs to be considered here, whether and to what extent they are helpful, is explained below in a way that is understandable for laypeople.
A brief summary of the scientific report for anyone interested in learning more about natural support for epilepsy.
A glossary, following the bibliography, explains technical terms.
The third part is aimed at medical professionals, presents the current state of pharmacological therapy, as well as study results regarding supportive essential oils in epilepsy, including source citation.
What is epilepsy?
The brain is made up of billions of nerve cells that constantly send electrical signals. Normally, this happens in an orderly fashion, like a well-conducted orchestra. In epilepsy, a kind of “electrical storm” suddenly occurs in the brain, called a seizure: many nerve cells fire simultaneously and uncontrollably.
What happens during a seizure?
Depending on which part of the brain is affected, a seizure can look very different:
- Absence
Brief “fading out” for seconds, the affected person stares into space - Myoclonic seizure
Short, twitching movements (often in the morning) - Focal seizure Twitches or sensations in a body part, sometimes with clouded consciousness
- Generalized tonic-clonic seizure (formerly “Grand Mal”): Fall, stiffening, rhythmic jerking of the entire body, unconsciousness
How common is epilepsy?
Epilepsy affects approximately 50 million people, ...which is about 1.1% of the world's population. In Germany, there are approximately 400,000–500,000 people living with epilepsy. It is the third most common neurological disorder after migraine and stroke.
The Big Problem
Around 30 % of the patients If medications are not sufficiently effective, it is referred to as drug-resistant epilepsy. These individuals often seek complementary options.
Epilepsy is a condition that affects the brain. It causes repeated seizures. Seizures happen when there is a sudden surge of electrical activity in the brain. This surge can disrupt the normal functioning of the brain. The electrical activity can spread from one area of the brain to another. This can cause a wide range of symptoms, depending on which part of the brain is affected.
Two systems must be in balance:
- GABA (Gamma-Aminobutyric Acid): The “Brake,” Calms the Brain
- GlutamateThe “Gas Pedal” Activates the Brain
In epilepsy, the balance is disturbed: too little brake, too much gas, the brain “goes haywire.”.
Furthermore, ion channels play an important role, which are tiny “gates” in the nerve cell membranes:
- Sodium channels (Nav)
When a nerve cell fires, in epilepsy they open too easily - Calcium channels (Cav)
Important for certain seizure types (e.g., absence seizures) - Potassium channels (Kv)
Help to calm the nerve cell after firing
How is epilepsy usually treated?
The most important antiepileptic drugs (AEDs)
Valproate (Valproic acid) is the most important broad-spectrum antiepileptic. It acts through multiple pathways simultaneously and is particularly effective for generalized epilepsies by inhibiting sodium and calcium channels and increasing GABA.
Important warning:
Valproate is highly harmful to unborn children, women of childbearing age!
Carbamazepine and Lamotrigine are the “door stoppers” for sodium channels. They prevent nerve cells from firing too easily. The two pharmaceuticals represent the first-line therapy for focal epilepsies. Lamotrigine is particularly well-tolerated and safe during pregnancy
Levetiracetam (Keppra) It has a unique mechanism, as it binds to a protein (SV2A) that regulates signal transmission between nerve cells. It is characterized by very good tolerability and has no interactions with other medications. Possible side effects include mood swings and irritability.
Topiramate It works in four different ways simultaneously—a “multi-talented” drug, so to speak—achieving a high seizure-free rate of 44–83% among patients. Possible side effects include slowed thinking and difficulty finding the right words.
Ethosuximide used for absence seizures (brief “drop attacks”). It blocks specific calcium channels in the thalamus (a relay center in the brain).
Phenobarbital and benzodiazepines enhance the GABA effect (the “brake”). They are cost-effective, effective, but have sedative and addictive potential (benzodiazepines).
Essential oils for epilepsy – what's possible?
Caution is advised with epilepsy!
Some essential oils can trigger or worsen seizures (proconvulsant). ALWAYS talk to your neurologist before using essential oils.
Oils to AVOID with Epilepsy:
- Rosemary (in high doses - camphor content)
- Eucalyptus (in high doses)
- Peppermint Pulegone content
- Fennel (Fennel in high doses)
- Sage (Thujone content)
- Camphor (directly proconvulsive)
Which essential oils show anticonvulsant properties?
Lavender – calming and anticonvulsant
Lavender (Lavandula angustifolia) is the best-researched oil for epilepsy.
composition
– Linalool (25–45 %)
– Linalyl acetate (25–50 %)
Effect
– Activates GABA-A receptors (the brain's “brake”)
– Inhibits sodium channels (prevents uncontrolled firing)
– Reduces seizure susceptibility
Clinical Effects
– Linalool significantly prolonged the time to the first seizure in animal experiments using the PTZ model (standard model for seizure tests).
– Some patients report that lavender aromatherapy delays seizures or alleviates auras (warning signs).
– Use in an aroma diffuser for inhalation, but never internally without medical advice
Rosemary/Borneol – neuroprotective (with caution!)
Borneol is an active ingredient from rosemary and other plants.
Effect
- Protects nerve cells from damage from repeated seizures; GABAergic and neuroprotective
Borneol reduced seizure severity in the kindling model and lowered inflammatory markers (GFAP) in animal experiments
Attention: Rosemary oil as a whole can be pro-convulsant in high doses, only take isolated borneol or very low doses
Oregano – Carvacrol and Thymol
Carvacrol (from oregano and thyme) shows interesting anticonvulsant properties.
Effect
– Inhibits sodium channels (like carbamazepine)
– Modulates TRPV1 receptors (heat and pain receptors)
Reduced seizure duration and severity in animal experiments
Eucalyptus - 1,8-Cineole (use with caution!)
1,8-Cineole Eucalyptus shows anticonvulsant effects in an animal model.
Effect
– Does the NO (nitric oxide) system in the brain influence
– Increases seizure threshold (~25 mg/kg vs. ~10 mg/kg in control group)
– Activates Nrf2 (antioxidant protection factor)
- Attention: In high doses proconvulsant, only in small amounts and after consultation with a doctor
Chamomile – Apigenin and Synergies
Camomile (Matricaria chamomillacontains apigenin, a flavonoid with anxiolytic and anticonvulsant properties.
Effect
– Apigenin binds to GABA-A receptors (at the same site as benzodiazepines, but much weaker)
– New study (2025): Combination of oregano + chamomile + lavender (administered intranasally) showed synergistic anticonvulsant effect in animal model – better than any oil alone
- As a supplement for mild seizures or for anxiety reduction
β-Caryophyllene - The Anti-Inflammatory
β-Caryophyllene (BCP) is found in black pepper, lavender, rosemary, and cannabis.
Effect
Activates CB2 receptors (cannabinoid receptors) without psychoactive effect
– Inhibits NF-κB (the inflammation switch)
– Reduces neuroinflammation, which is important because inflammation can worsen epilepsy
BCP is also found in hemp oil and could be part of the effect of CBD-rich extracts
The most important plant Cannabis and CBD
CBD (Cannabidiol) – The Clinical Breakthrough
CBD is an active ingredient from the cannabis plantCannabis sativa), but without the well-known psychoactive effects of THC. It is the only plant-based antiepileptic drug officially approved by the FDA (USA) and EMA (Europe) under the name Epidiolex was approved.
It is administered orally as a prescription medication (Epidiolex) in the form of an oil solution twice daily.
Effect
– Inhibits sodium channels (like carbamazepine)
– Modulates TRPV1 and GPR55 (specialized receptors)
– Anti-inflammatory via CB2 receptors – Protects nerve cells
Side effects
– Drowsiness, diarrhea, decreased appetite
– Can increase the levels of other antiepileptics (especially clobazam), dose adjustment necessary
Authorization for
- Dravet Syndrome A severe form of epilepsy that begins in infancy
- Lennox-Gastaut Syndrome (LGS) Severe epilepsy with multiple seizure types
Studies
– In Dravet syndrome: CBD reduced seizure frequency by 38.9% compared to 13.3% with placebo, 66.7% of patients (1-year and 3-year survival rates) had ≥ 50 fewer % seizures (p = 0.009)
– In the LGS trial: 43.9% reduction in falls (%) compared with 21.8% (%) with placebo (p < 0.001)
– For other drug-resistant forms of epilepsy: Various studies show a 40–50% reduction in seizures
Comparison table - What helps how?
Safety rule
Possibly helpful (after consulting with a doctor):
- Lavender Aromatherapy (Diffuser, Inhalation) – relaxing, potentially seizure-reducing
- CBD (as Epidiolex) - for Dravet/LGS, prescription required
- β-Caryophyllene – anti-inflammatory, well-tolerated
- Chamomile – calming, anxiolytic
When you have epilepsy, AVOID:
- Rosemary oil (ingestion, high doses) – Camphor can trigger seizures
- Eucalyptus oil (high doses)
- Peppermint oil (high doses, pulegone)
- Fennel oil Fennel
- Sage oil Thujone
- Camphor oil (directly proconvulsive)
General Safety Instructions
- ALWAYS speak with the neurologist first Epilepsy is serious
- Never stop taking antiepileptic drugs on your own. – can lead to life-threatening status epilepticus
- Consider interactions: Some oils can alter the levels of anti-epileptic drugs.
- Do not put on the skin – always dilute
- Children: Special caution, doses much lower
Frequently Asked Questions
Can I take lavender instead of my anti-epileptic medication?
No, absolutely not! Lavender has only shown anticonvulsant effects in animal studies. Stopping antiepileptic drugs on your own can be life-threatening.
What about CBD oil from the internet?
CBD products from the internet are not the same as the approved medication Epidiolex. They are not standardized, may contain other cannabinoids, and have no proven efficacy. Talk to your neurologist about prescription CBD.
Can aromatherapy stop a seizure?
There are reports that some patients can delay seizures through certain smells (like lavender), but this is not a reliable method and not a substitute for medication.
Are essential oils safe for children with epilepsy?
Only after express consultation with the pediatric neurologist. Children react more sensitively, and some oils are dangerous for children.
Do I have to buy expensive oils?
Quality is important: Look for 100% % pure essential oils, ideally with a batch-specific analysis certificate (GC/MS).
Inexpensive perfume oils or synthetic fragrances have no therapeutic effect and, due to their synthetic ingredients, can be harmful to health and cause headaches, nausea, etc.
Anyone who wants to learn more about the selection and quality of essential oils will find information in the article „Essential Oils - Odyssey of a Search“found.
Another contribution quotes Prof. Dr. Dr. Dr. med. habil. Hanns Hatt from the Ruhr University Bochum, who in his video „Healing with fragrances“explains the effect of essential oils on the human body in an interesting, entertaining, and yet scientific way.
The document provides a summary in three sentences. It offers a concise overview of the main points discussed. This allows for a quick understanding of the content without needing to read the full text.
Epilepsy is a serious neurological disorder in which electrical disturbances in the brain lead to seizures, and for 30% of patients, standard medications are not sufficiently effective.
Essential oils and terpenes, especially linalool (lavender), borneol, and carvacrol, show anticonvulsant properties in animal studies through GABA-A activation and sodium channel inhibition; CBD (cannabidiol) is the only plant-based antiepileptic with clinical approval (Dravet syndrome, Lennox-Gastaut syndrome).
Essential oils can be used as a supplement for epilepsy, but only after explicit consultation with the treating neurologist, as some oils can also trigger seizures.
This text is based on the scientific report “Epilepsy and Essential Oils: Pharmacology, Mechanisms of Action, and Complementary Therapeutic Approaches” and is for general informational purposes only. Epilepsy is a serious illness; always consult your neurologist for questions regarding treatment.
Recommended dōTERRA- Products for adjunctive therapy in epilepsy
Potentially safe products (based on study findings)
Oils to AVOID with epilepsy
DIY Blends
Blend 1 – “Neuroprotective” (Diffuser)
Target: Neuroprotective protection, GABA modulation
Application: 30 minutes daily in the diffuser, especially in the evening
Blend 2 – “Aura Management” (Topical, Fast)
Target: For aura symptoms: Attempt seizure interruption (as a supplement ONLY, not a substitute for emergency medication)
Application: On temples, neck, wrists – immediately at the start of an aura
Emergency medication (e.g., diazepam rectal) remains a priority
Blend 3 – “Anxiety & Stress Relief” (Diffuser/Topical)
Target: Stress ↓ (more frequent seizure trigger), Relaxation ↑
Application: During stress or before known trigger situations
Blend 4 - “Sleep & Relaxation” (Evening)
Target: Sleep quality ↑ (Sleep deprivation = seizure trigger)
Application: In the diffuser, 30 minutes before sleep
Blend 5 – “Anti-Inflammation” (Topical)
Target: Neuroinflammation ↓ (Epilepsy Pathophysiology)
Application: Massage temples, neck, and spine
Application Protocol - 4-Week Plan
Week 1 - Cautious Start
- Consult a doctor Before starting with oils
- In the evening Blend 4 in diffuser (sleep)
- Daily: Blend 1 in diffuser (30 min)
- Seizure log run (date, time, trigger)
Week 2 – Expansion
- Mornings: Blend 3 in stressful situations
- In the evening Blend 4 (Sleep) + Blend 5 topical
- Blend 2: Prepare for Aura situations
Week 3-4 – Optimization
- Evaluate seizure record has anything changed?
- Neurologists inform About oil application
- Adaptation based on individual tolerance
Combination with others dōTERRA-The product
Emergency plan for seizure
- Security Person in recovery position
- Measure time: Seizure lasting longer than 5 minutes → Emergency doctor (112)
- Emergency medication Diazepam-Rectal / Midazolam-Nasal (as prescribed by doctor)
- NO OIL in the mouth or nose during an active seizure
- Essential oils are NOT emergency medication
Epilepsy and Essential Oils – Pharmacology, Mechanisms of Action, and Complementary Therapeutic Approaches
A Comprehensive Scientific Report on Standard Antiepileptic Drugs, Terpenes, and Essential Oils – Molecular Basis, Clinical Evidence, and Adjuvant Therapy Strategies
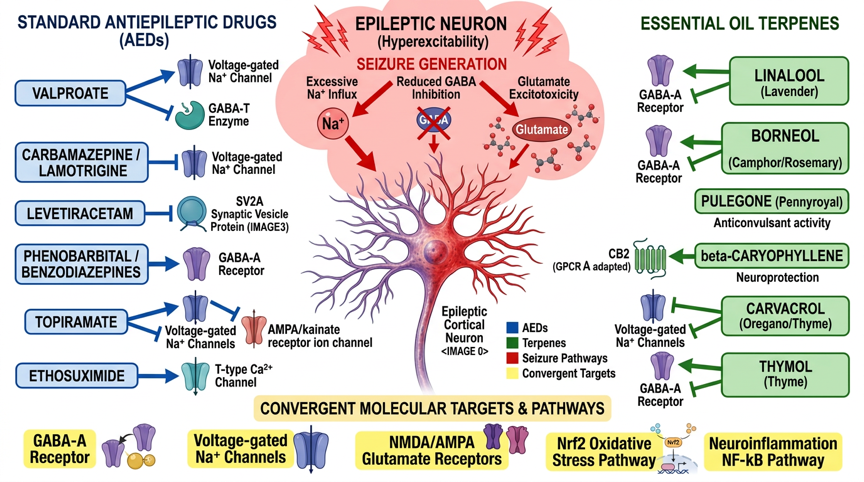
Fig. 1: Molecular signaling pathways of standard antiepileptic drugs and essential oils in epilepsy – Standard AEDs (Valproate/Nav+GABA-T, Carbamazepine/Nav, Levetiracetam/SV2A, Topiramate/multimodal, Ethosuximide/T-type Ca2+, Phenobarbital/GABA-A) and terpenes (Linalool/GABA-A+Nav, Borneol/GABA-A+Nav, Carvacrol/Nav+TRPV1, β-Caryophyllene/CB2, 1,8-Cineole/NO+Nrf2) with convergent target molecules (GABA-A receptor, Nav channels, NMDA/AMPA glutamate, NF-κB neuroinflammation, Nrf2 oxidative stress)
Top: Sharp wave (70-200 ms) followed by slow wave.
Content: 3-Hz Spike-Wave Complex (GSWD) - Hallmark Pattern of Absence Epilepsy.
Below: Polyspike complexes (3-5 spikes, 4-5 Hz) – characteristic of Juvenile Myoclonic Epilepsy (JME).
Introduction
Epilepsy is one of the most common chronic neurological disorders worldwide and affects an estimated 50 million people, or about 1% of the global population [D1]. It is defined by a persistent predisposition to epileptic seizures as a result of pathologically synchronized neuronal discharges, as well as by the neurobiological, cognitive, psychological, and social consequences of this condition. [D2]. Despite significant advances in the development of antiepileptic drugs (AEDs), approximately 30% of patients continue to suffer from drug-resistant epilepsy, in which two or more AEDs used at appropriate doses fail to achieve adequate seizure control [D3].
Standard treatment for epilepsy includes stepwise pharmacotherapy with first-, second-, and third-generation antiepileptic drugs. Valproate, carbamazepine, and lamotrigine are considered first-line medications for focal and generalized epilepsies; newer substances such as levetiracetam, topiramate, lacosamide, and perampanel expand the therapeutic arsenal. [D4]. Despite their effectiveness, AEDs are associated with significant side effects, including cognitive impairment, teratogenicity (valproate), bone marrow depression (carbamazepine), weight gain, and psychiatric disorders, which significantly impact patients' quality of life. [D5].
Against this backdrop, scientific interest in plant-based active compounds and essential oils as adjunctive or alternative therapy options is growing. Numerous terpenes and bioactive compounds demonstrate anticonvulsant, neuroprotective, anti-neuroinflammatory, and antioxidant properties in preclinical and clinical studies, often through mechanisms that are complementary to classic AEDs. [D6]. Cannabidiol (CBD), a plant-based active compound derived from *Cannabis sativa*, has already received clinical approval for specific epilepsy syndromes and demonstrates the potential of botanical compounds in epilepsy therapy. [D7]. This report systematically analyzes the pharmacology of standard antiepileptic drugs, the molecular mechanisms of action of relevant essential oils and terpenes, and the available clinical evidence for their adjuvant use in epilepsy.
EEG in Epilepsy – Fundamentals and Interpretation Principles
The electroencephalogram (EEG) is the central diagnostic tool for epilepsy and allows for the direct measurement of cortical electrical activity via scalp electrodes. It is used for seizure classification, lesion localization, and therapy monitoring.
Interictal Phase: The EEG between seizures typically shows interictal epileptiform discharges (IEDs): spikes (<70 ms), sharp waves (70-200 ms), and 3-Hz spike-wave complexes (GSWDs in absence epilepsy). These IEDs arise from synchronous neuronal discharges in the epileptic focus.
Ictal Phase: The ictal EEG frequently begins with low-voltage fast activity (LVFA, 20-35 Hz) as the onset pattern. Over time, a rhythmic theta-delta evolution with increasing amplitude develops. Termination is abrupt, followed by postictal suppression.
Postictal phase: After seizure termination, the EEG shows suppression (marked amplitude reduction) followed by diffuse delta slowing (0.5–3.5 Hz). Recovery to normal background activity occurs within minutes to hours, depending on seizure duration and type.
Effect of β-caryophyllene on the epileptic EEG: β-caryophyllene (BCP), a sesquiterpenoid CB2 agonist found in essential oils (e.g., black pepper, cloves), reduces seizure frequency by 66% in the PTZ model, increases seizure latency by 187% and lowers spike frequency by 27–68%. The EEG shows a normalization of spectral power: gamma power (a marker of hyperexcitability) decreases, while delta power increases. The mechanism of action involves CB2 agonism (reducing neuroinflammation) and GABA-A modulation (increasing chloride influx).
Pathophysiology of Epilepsy
Neuronal Hyperexcitability and Synchronization
The fundamental pathophysiology of epilepsy is based on an imbalance between excitatory and inhibitory neurotransmission, which leads to paroxysmal synchronized neuronal discharge. At the cellular level, an epileptic seizure arises from the interplay of ion channel dysfunctions, disturbed synaptic transmission, and altered network activity. [D8]. Mutations or functional changes in voltage-gated sodium (Nav), potassium (Kv), and calcium (Cav) channels, as well as ligand-gated ion channels (GABA-A, NMDA), contribute to increased neuronal excitability. [D9].
Above (blue): Interictal phase with IEDs (Spikes <70 ms, Sharp Waves 70-200 ms, 3-Hz GSWDs).
Middle (red): Ictal Phase – LVFA-Onset (20-35 Hz), Theta-Delta Evolution, abrupt termination.
Below (purple): Postictal phase – EEG suppression, diffuse delta slowing (0.5-3.5 Hz)
GABA/Glutamate Imbalance
The balance between the inhibitory neurotransmitter GABA (gamma-aminobutyric acid) and the excitatory neurotransmitter glutamate is crucial for the seizure threshold. Reduced GABAergic inhibition, due to decreased GABA synthesis, reduced GABA-A receptor density, or increased GABA transaminase activity, lowers the seizure threshold. [D10]. Simultaneously, excessive activation of NMDA and AMPA glutamate receptors promotes depolarization spread and epileptic discharges. This GABA/glutamate imbalance is the central pharmacological target of most AEDs. [D11].
Ion channel dysfunction
Voltage-gated sodium channels (Nav1.1, Nav1.2, Nav1.6) play a key role in action potential generation and propagation. Gain-of-function mutations in Nav1.1 (Dravet syndrome) or Nav1.2 lead to uncontrolled discharges. [D12]. T-type calcium channels (Cav3.1, Cav3.2) are central to absence epilepsies: their rhythmic activation in the thalamus generates the characteristic 3-Hz spike-wave discharges. [D13]. Potassium channel defects (KCNQ2/3, KCNT1) affect repolarization and increase seizure susceptibility.
Neuroinflammation and oxidative stress
Chronic epilepsy is associated with persistent neuroinflammation: Activated microglia and astrocytes release pro-inflammatory cytokines (IL-1β, IL-6, TNF-α), which destabilize the blood-brain barrier and increase neuronal excitability. [D14]. The transcription factor NF-κB coordinates this neuroinflammatory cascade. Concurrently, repetitive seizures lead to oxidative stress through mitochondrial dysfunction and ROS production, which promotes neuronal apoptosis and hippocampal sclerosis. [D15]. The Nrf2 signaling pathway, as the master regulator of the antioxidant response, is often suppressed in drug-resistant epilepsy.
Drug resistance mechanisms
In 30% of epilepsy patients, two or more AEDs fail. Drug resistance arises from: (1) overexpression of P-glycoprotein (P-gp/MDR1) and other ABC transporters that efflux AEDs from the brain; (2) alterations in AED target structures (e.g., Nav channel mutations that reduce phenytoin binding); (3) pharmacogenetic variants in CYP metabolism; (4) neuroinflammation, which alters blood-brain barrier permeability [D16].
Pharmacology of Standard Antiepileptic Drugs
Sodium channel blockers – Carbamazepine, Lamotrigine, Phenytoin, Lacosamide
Sodium channel blockers are the most common and oldest class of AEDs. They bind to the inactivated state of voltage-gated sodium channels (Nav1.1–Nav1.6) and prolong their inactivation duration, the principle of “use-dependent blockade”.” [D8]. Carbamazepine (CBZ) and oxcarbazepine are first-line therapies for focal epilepsies; lamotrigine additionally inhibits presynaptic glutamate release and is particularly effective in Lennox-Gastaut syndrome and absence epilepsy. [D17]. Phenytoin/Fosphenytoin stabilizes the inactivated Nav channel state and is a standard medication in status epilepticus. Lacosamide (newer generation) enhances “slow inactivation” of Nav channels via a unique mechanism and demonstrates favorable pharmacokinetics. [D18].
Clinical efficacy: Carbamazepine achieves seizure freedom in 40–50% of patients with focal epilepsy when used as monotherapy; lamotrigine demonstrates comparable efficacy with a better tolerability profile, particularly in women of childbearing age [D4].
Valproate - Broad-spectrum antiepileptic
Valproic acid (valproate/VPA) is the most important broad-spectrum antiepileptic drug with multiple mechanisms of action: inhibition of voltage-gated sodium channels, reduction of T-type calcium currents, inhibition of GABA transaminase (GABA-T) with consecutive GABA increase, and activation of glutamate decarboxylase (GAD). [D19]. Valproate is the first-line treatment for generalized epilepsy (juvenile myoclonic epilepsy, absence epilepsy, Lennox-Gastaut syndrome) and achieves seizure freedom in 50–70% of patients with idiopathic generalized epilepsy [D4]. Critical: Valproate is highly teratogenic (neural tube defects, cognitive impairments in newborns) and should only be used in women of childbearing potential after careful risk-benefit assessment. [D5].
Levetiracetam – SV2A Binding
Levetiracetam (LEV) has a unique mechanism of action: it selectively binds to the synaptic vesicle protein 2A (SV2A), which regulates neurotransmitter exocytosis. D20. Through SV2A binding, LEV reduces presynaptic neurotransmitter release at high-frequency discharges, a “frequency-dependent” mechanism that barely affects normal synaptic transmission. LEV is first-line therapy for focal and generalized epilepsies, exhibits excellent pharmacokinetics (no enzyme induction, no protein binding), and is relatively safe during pregnancy. D20. Important side effect: behavioral changes (irritability, depression) in up to 15% of patients.
Topiramate – Multimodal Mechanism
Topiramate (TPM) is a multimodal AED with at least four mechanisms of action:
- Blockade of voltage-gated sodium channels,
- Enhancement of GABAergic inhibition at GABA-A receptors
- Antagonism at AMPA/Kainate Glutamate Receptors
- Inhibition of carbonic anhydrase (CA-II, CA-IV) [D21]
Clinical studies show seizure-free rates of 44–83% (6–7 months) and 41–76% (12–13 months) in monotherapy comparative trials; topiramate is as effective as valproate and carbamazepine [D21]. Important side effects: cognitive impairment (“word-finding difficulties”), weight loss, kidney stones.
GABA-A Modulators - Phenobarbital, Benzodiazepines, Vigabatrin
Phenobarbital (barbiturate) and benzodiazepines (diazepam, clonazepam, clobazam) enhance GABA-A receptor-mediated chloride inhibition: barbiturates prolong the opening duration of the chloride channel, benzodiazepines increase the opening frequency. [D22]. Phenobarbital is one of the oldest and cheapest AEDs, is frequently used in developing countries, but has significant sedation potential. Vigabatrin irreversibly inhibits GABA transaminase (GABA-T), thereby increasing GABA concentration in the brain; it is effective in infantile spasms but is associated with irreversible visual field deficits. [D23].
Ethosuximide – T-type calcium channel blocker
Ethosuximide (ESM) is a selective blocker of T-type calcium channels (Cav3.1/3.2) and a first-line therapy for childhood absence epilepsy. [D13]. T-type channels generate the rhythmic thalamic discharges that characterize absence seizures; blocking them effectively interrupts this cycle. Ethosuximide is as effective as valproate in treating absence epilepsy (seizure freedom ~50–60%), but has fewer side effects [D13].
Newer AEDs – Lacosamide, Perampanel, Brivaracetam
Lacosamide (LCM) selectively enhances the “slow inactivation” of voltage-gated sodium channels and inhibits Collapsein-Response-Mediator-Protein 2 (CRMP-2). [D18]. Perampanel (PER) is the first selective, non-competitive AMPA glutamate receptor antagonist and is particularly effective in drug-resistant generalized tonic-clonic seizures. [D24]. Brivaracetam (BRV) binds to SV2A like levetiracetam, but with 10 times higher affinity and faster onset of action. [D25].
Essential Oils as Adjuvant Therapy - Mechanistic Basis
Pharmacokinetics and CNS Penetration
Terpenes from essential oils are characterized by high lipophilicity and low molecular weight (< 300 Da), which allows for efficient penetration of the blood-brain barrier (BBB). [D26]. When inhaled, volatile terpenes are transported via the olfactory mucosa directly into the olfactory bulb and the limbic system, a direct pathway that bypasses the blood-brain barrier and leads to measurable CNS concentrations within minutes. [D27]. Transdermal and oral application allow for systemic absorption; oral bioavailability varies greatly depending on the terpene and formulation.
Anticonvulsant mechanisms of action
Essential oils and their terpenes can act through multiple anticonvulsant mechanisms that overlap with those of classical AEDs:
- Modulation of voltage-dependent sodium channels (Linalool, Borneol, Carvacrol, Thymol)
- Enhancement of GABAergic inhibition at GABA-A receptors (linalool, borneol, thymol, apigenin)
- Inhibition of glutamatergic excitation at NMDA/AMPA receptors
- Antioxidative neuroprotection via Nrf2 activation; (5) Anti-neuroinflammatory effects via NF-κB inhibition (β-caryophyllene) [D28].
Neuroprotective properties
Many terpenes show neuroprotective properties that go beyond direct anticonvulsant effects. By inhibiting apoptosis signaling pathways (caspase-3, Bax/Bcl-2 ratio), reducing lipid peroxidation (MDA levels), and increasing antioxidant enzyme activities (SOD, CAT, GPx), terpenes protect against seizure-induced neuronal damage. [D29]. These neuroprotective properties are particularly relevant in drug-resistant epilepsy, where repetitive seizures lead to progressive brain damage.
Complementary Mechanisms of Action to Standard Antiepileptic Drugs
The following table shows the complementary mechanisms of action of essential oils compared to standard antiepileptic drugs at a molecular level:

Standard-AEDs (Valproate/Nav+GABA-T, Carbamazepine/Nav, Levetiracetam/SV2A, Topiramate/multimodal, Ethosuximide/T-type Ca²⁺, Phenobarbital/GABA-A) and Terpenes (Linalool/GABA-A+Nav, Borneol/GABA-A+Nav, Carvacrol/Nav+TRPV1, β-Caryophyllene/CB2, 1,8-Cineole/NO+Nrf2) with convergent molecular targets (GABA-A, Nav, NMDA/AMPA, NF-κB, Nrf2/ARE).
Specific essential oils and clinical evidence
Lavender Oil (Lavandula angustifolia) – Linalool
Lavender oil is the most extensively studied essential oil in the context of neurological disorders. The main active ingredient, linalool (25–45 %), exhibits anticonvulsant properties in preclinical studies: In a pentylentetrazol (PTZ) model, linalool inhalation increased the seizure threshold, prolonged the latency to the first seizure, and reduced seizure duration [D30]. Mechanistically, linalool inhibits voltage-gated sodium channels by increasing the action potential threshold and reducing the upstroke velocity, as well as by modulating calcium-activated potassium currents. [D31]. A pilot study on olfactory training in patients with epilepsy (n=11) used lavender as an olfactory conditioning stimulus and showed a reduction in seizures of ≥50% in 6 of 11 patients, although the authors note the limited data available [D32].
In a study with several essential oils in a PTZ mouse model, *Lavandula angustifolia* showed a significant increase in seizure latency (p < 0.05) and reduction in seizure severity compared to the control group. [D33]. Silexan (80 mg orally, standardized lavender extract) is clinically validated for anxiety disorders and could be used adjunctively for epilepsy-associated anxiety. [D34].
Cannabis oil / Cannabidiol (CBD)
Cannabidiol (CBD) is the best-known plant-based active ingredient with clinically proven anticonvulsant effects. As a non-psychoactive phytocannabinoid derived from Cannabis sativa, CBD has multiple anticonvulsant mechanisms: antagonism of GPR55 (lyso-phosphatidylinositol receptor), modulation of TRPV1 channels, inhibition of voltage-gated sodium channels, and modulation of the endocannabinoid system. [D35]. Pharmaceutical CBD (Epidiolex®) is FDA (2018) and EMA (2019) approved for Dravet syndrome and Lennox-Gastaut syndrome [D7].
Clinical evidence: In placebo-controlled Phase III trials in patients with Dravet syndrome (n=120), CBD (20 mg/kg/day) reduced the monthly frequency of convulsive seizures by 38.9% compared with 13.3% for placebo (p = 0.01) [D7]. In Lennox-Gastaut syndrome (n=225), CBD reduced drop attacks by 43.9 % compared with 21.8 % in the placebo group (p < 0.001) [D36]. In a triple-blind study of refractory frontal lobe epilepsy (n=27), 66.7% of patients receiving CBD showed improvement after 4 weeks, compared with 20.0% of those receiving placebo (mean difference 45.58; 95% CI: 8.99–82.18; p = 0.009) [D37].
Rosemary oil (Rosmarinus officinalis) - Borneol, 1,8-Cineole
Rosemary oil contains borneol (up to 20%), 1,8-cineole/eucalyptol (35–50%), camphor (10–20%), and α-pinene [D38]. Borneol exhibits antiepileptogenic effects in a PTZ kindling model: it reduces seizure progression, lowers GFAP markers (neuroinflammation) in the hippocampus, reduces oxidative stress, and modulates GABAergic transmission. [D39]. In a PTZ threshold test, 1,8-cineole increased the seizure threshold (~25 mg/kg vs. ~10 mg/kg in control) and reduced malondialdehyde (MDA) levels as a marker for oxidative stress. [D40]. In the PTZ multi-oil study, Rosmarinus officinalis showed a significant prolongation of seizure latency and reduction in seizure severity. [D33].
Oregano/Thyme Oil - Carvacrol and Thymol
Carvacrol (oregano, thyme, savory) and thymol (thyme) are structurally related monoterpenoid phenols with demonstrated anticonvulsant activity. D41. Carvacrol inhibits voltage-gated sodium channels (Nav1.2, Nav1.4) and TRPV1 channels, enhances GABAergic inhibition, and shows anticonvulsant effects in PTZ and kainic acid models. [D42]. Thymol positively allosterically modulates GABA-A receptors (similar to barbiturates) and inhibits voltage-dependent sodium channels. [D43]. Important caveat: In an animal study, Origanum vulgare oil exhibited toxic effects at the tested concentration (100% mortality), underscoring the importance of standardized dosing and formulation [D33].
Eucalyptus Oil (Eucalyptus globulus) – 1,8-Cineole
Eucalyptus oil contains 60–90% 1,8-cineole (eucalyptol), a monoterpene oxide with anticonvulsant, anti-inflammatory, and antioxidant properties [D44]. 1,8-Cineole modulates the nitric oxide (NO) system, which plays a complex, bidirectional role in seizure regulation: low NO levels can be anticonvulsant, while high NO levels can be proconvulsant. In a PTZ model, 1,8-cineole significantly increased the seizure threshold and reduced lipid peroxidation. [D40]. Eucalyptus oil inhalation also shows anti-neuroinflammatory effects through NF-κB inhibition and reduction of pro-inflammatory cytokines. [D45].
Frankincense Oil (Boswellia sacra) - α-Pinene, Boswellic Acids
Frankincense oil contains α-pinene (up to 75%), limonene, and non-volatile boswellic acids [D46]. α-Pinene inhibits acetylcholinesterase and has anxiolytic properties; Boswellic acids (AKBA) inhibit 5-lipoxygenase and NF-κB, reducing neuroinflammatory processes in epilepsy. In animal models, Boswellia extract showed neuroprotective effects after kainic acid-induced seizures: reduction of hippocampal neuron loss, decreased GFAP expression, and reduced IL-1β levels. [D47]. Incensol acetate from frankincense activates TRPV3 channels and shows anxiolytic effects independently of GABA-A or opioid receptors.
Chamomile Oil (Matricaria chamomilla) – Apigenin, α-Bisabolol
Chamomile oil contains α-bisabolol (up to 50%), chamazulene, and the flavonoid apigenin [D48]. Apigenin binds to the benzodiazepine binding site of the GABA-A receptor (Ki = 4 µM) and exhibits anxiolytic and anticonvulsant properties without sedation in preclinical models. [D49]. In a synergy study (2025), the combination of oregano, chamomile, and lavender (intranasal) showed synergistic anticonvulsant activity in the PTZ model, with a significant reduction in seizure frequency (p < 0.05) and optimized effects at combined lower doses compared to individual substances. [D50].
Fennel oil (Foeniculum vulgare) – trans-Anethole
Fennel oil contains trans-anethol (50–80 %), fenchone, and estragole. Trans-anethol modulates GABA-A receptors and inhibits NMDA glutamate receptors, which explains its anticonvulsant effects in animal models [D28]. However, fenchone (a component of fennel oil) has proconvulsant properties and can trigger seizures, which underscores the importance of standardized, low-fenchone fennel oil preparations for therapeutic applications.
Molecular Mechanisms of Terpenes in Epilepsy
Linalool – Sodium Channel Inhibition and GABA-A Modulation
Linalool (3,7-Dimethyl-1,6-octadien-3-ol), the main monoterpene in lavender and coriander, exhibits dose-dependent effects on neuronal excitability. In isolated neurons, linalool increases the action potential threshold and reduces the upstroke velocity, both indicators of sodium channel blockade. [D31]. Simultaneously, linalool activates calcium-dependent potassium channels (BK channels), which accelerate repolarization and reduce firing frequency. Linalool's GABA-A modulation increases chloride-mediated inhibition. [D30]. Important: At high doses, linalool can have a paradoxical stimulating effect, emphasizing the importance of dose optimization.
Borneol – GABAergic Modulation and Neuroprotection
Borneol (2-Bornanol), a bicyclic monoterpene alcohol found in camphor trees and rosemary, exhibits antiepileptogenic effects in the PTZ kindling model. It modulates GABAergic transmission, reduces oxidative stress (decreased MDA levels, increased SOD activity), and lowers neuroinflammatory markers (GFAP) in the hippocampus. [D39]. Borneol also inhibits voltage-gated sodium channels and shows synergistic anticonvulsant effects in combination with standard AEDs in animal models.
Carvacrol and Thymol – Nav Channel Blockade and GABA-A Modulation
Carvacrol (5-Isopropyl-2-methylphenol) and thymol (2-Isopropyl-5-methylphenol) are structural isomers of monoterpene phenols with similar, but not identical, mechanisms of action. Carvacrol inhibits Nav1.2 and Nav1.4 sodium channels, as well as TRPV1 channels, and exhibits neuroprotective effects in kainic acid models. [D42]. Thymol positively allosterically modulates GABA-A receptors at a binding site similar to barbiturates and inhibits voltage-gated sodium channels, a dual mechanism resembling that of phenobarbital. [D43].
β-Caryophyllene – CB2 Agonism and Anti-Neuroinflammation
β-Caryophyllene (BCP), the bicyclic sesquiterpene found in black pepper and cloves, acts as a selective CB2 cannabinoid receptor agonist and has demonstrated anticonvulsant and neuroprotective effects in animal models. [D51]. The anticonvulsant effects of BCP are partly mediated via GABAergic, serotonergic, and nitrergic systems. Inhibition of NF-κB by BCP reduces neuroinflammatory processes that contribute to epileptogenesis. In a mouse model, BCP significantly reduced PTZ-induced seizure frequency and demonstrated neuroprotective effects in the hippocampus. [D52].
and effects in the PTZ model and mechanism of action
A: Control - 3 seizures in 20 s, high spike frequency
B: BCP 200 mg/kg — 1 seizure (↓66 %), latency ↑187 %, spikes ↓68 %.
C: Spike frequency is dose-dependent (↓27–68 %).
Spectral Power — Gamma Power ↓, Delta Power ↑ (Normalization).
E: Mechanism of action — BCP → CB2 (↓Neuroinflammation) + GABA-A (↑Chloride influx → ↓Hyperexcitability).
1,8-Cineole - NO System and Antioxidative Neuroprotection
1,8-Cineole (Eucalyptol), the main monoterpene in eucalyptus and rosemary oil, modulates the nitric oxide (NO) system and exhibits antioxidant neuroprotection [D40]. In a PTZ threshold test, 1,8-cineole significantly increased the seizure threshold (~25 mg/kg vs. ~10 mg/kg control group) and reduced malondialdehyde (MDA) levels as a marker of lipid peroxidation. Additionally, 1,8-cineole inhibits NF-κB-dependent neuroinflammatory processes and activates the Nrf2 signaling pathway for antioxidative neuroprotection.
New and supplementary essential oils
Lemon balm oil (Melissa officinalis) Rosmarinic acid
Lemon balm oil contains rosmarinic acid, citral, citronellal, and geraniol. [D53]. Rosemary acid inhibits GABA transaminase (GABA-T), the same mechanism as vigabatrin, thereby increasing GABAergic inhibition. Additionally, rosemary acid exhibits antioxidant and anti-neuroinflammatory properties by inhibiting COX-2 and NF-κB. In animal models, lemon balm extract showed anticonvulsant effects in PTZ-induced seizures.
Peppermint oil (Mentha piperita) – Menthol, pulegone
Peppermint oil contains menthol (30–55%), menthone (14–32%), and pulegone (<1%). In the PTZ multi-oil study, Mentha piperita showed no seizures at the tested concentration and 100% survival, the best safety profile of all tested oils [D33]. Menthol activates TRPM8 cold channels and modulates sodium channels; it exhibits analgesic and slightly anticonvulsant properties. Important: Pulegone in high concentrations is hepatotoxic and pro-convulsant.
Black Cumin Seed Oil (Nigella sativa) – Thymoquinone
Black cumin oil contains thymoquinone (TQ, 30–48 %), which exhibits potent anticonvulsant, neuroprotective, and anti-neuroinflammatory properties D54. TQ inhibits NMDA glutamate receptors, activates GABA-A receptors, and has shown efficacy comparable to valproate in animal models for PTZ-induced seizures. TQ activates Nrf2 and inhibits NF-κB, reducing oxidative stress and neuroinflammation in epilepsy.
Hemp Extract – CBD and Other Cannabinoids
In addition to pharmaceutical CBD, Cannabis sativa contains other anticonvulsant cannabinoids: CBDV (Cannabidivarin) has shown efficacy in clinical studies for focal seizures; THCV (Tetrahydrocannabivarin) possesses anticonvulsant properties in animal models; CBG (Cannabigerol) exhibits neuroprotective effects. [D55]. The entourage effect describes synergistic effects between different cannabinoids and terpenes in full-spectrum cannabis extract.
Clinical Evidence Compared to Standard Therapy
Randomized controlled trials (RCTs)
With the exception of pharmaceutical CBD, the clinical evidence for essential oils in epilepsy is predominantly preclinical. CBD has demonstrated convincing efficacy in several Phase III RCTs for Dravet syndrome and Lennox-Gastaut syndrome: a 38.9% reduction in convulsive seizures (Dravet, p = 0.01) and a 43.9% reduction in drop attacks (LGS, p < 0.001) [D7] [D36]. In a triple-blind study of refractory frontal lobe epilepsy (n=27), CBD demonstrated significant superiority over placebo (66.7 % vs. 20.0 % responders, p = 0.009) [D37].
Preclinical animal models
In a PTZ mouse model with multiple essential oils (Lavandula angustifolia, Rosmarinus officinalis, Mentha piperita, Origanum majorana), all tested oils (except Origanum vulgare) showed significant increases in seizure latency and reductions in seizure severity compared to the control group (p < 0.05). [D33]. A synergy study (2025) with combined intranasal application of oregano, chamomile, and lavender oils demonstrated synergistic anticonvulsant effects that went beyond the sum of the individual effects. [D50].
Olfactory training for epilepsy
A pilot study on olfactory training (Seizure Alert Aromatherapy) in epilepsy patients (n=11) used conditioned scent stimuli (lavender, jasmine) for seizure prediction and control. [D32]. Six out of 11 patients showed a ≥50% reduction in seizures. The principle is based on the conditioning of olfactory-limbic connections, which may have a seizure-modulating effect.
Comparison Table – Essential Oils vs. Standard Antiepileptic Drugs
Joint conclusion
Epilepsy is a complex neurobiological disorder that requires precise, individualized pharmacotherapy. The present analysis shows that essential oils and their terpenes exhibit complementary mechanisms of action to standard antiepileptic drugs and possess clinically relevant potential in specific areas, particularly neuroprotection, neuroinflammation, and adjuvant seizure modulation.
As a plant-based active ingredient, cannabidiol (CBD) has taken the crucial step from preclinical trials to clinical approval, demonstrating that botanical compounds can be effective in treating drug-resistant epilepsy. The approval of Epidiolex® for Dravet syndrome and Lennox-Gastaut syndrome, with seizure reductions of 38.9–43.9% (p < 0.001), represents a paradigm shift in epilepsy treatment.
For other essential oils, lavender (linalool), rosemary (borneol), eucalyptus (1,8-cineole), chamomile (apigenin), and black cumin (thymoquinone) have a solid preclinical evidence base with consistent anticonvulsant, neuroprotective, and anti-neuroinflammatory effects in animal models. The synergism study (2025) with combined intranasal application of oregano, chamomile, and lavender shows that combined formulations can achieve synergistic effects that go beyond individual substances.
For clinical practice, an integrated approach is recommended: standard AEDs as basic therapy in proven epilepsy, supplemented by adjuvant essential oils (especially CBD for pharmacoresistance, lavender for anxiety/sleep disorders, borneol/1,8-cineole for neuroprotection) under medical supervision. Future research must conduct standardized clinical trials with defined oil preparations, clear epilepsy diagnostic criteria, and validated endpoints (seizure frequency, severity, quality of life, biomarkers).
credentials
[D1] Fiest, K. M., Sauro, K. M., Wiebe, S., et al. (2017). Prevalence and incidence of epilepsy: A systematic review and meta-analysis of international studies. Neurology, 88(3), 296–303. https://doi.org/10.1212/WNL.0000000000003509
Fisher, R. S., Acevedo, C., Arzimanoglou, A., & et al. (2014). ILAE official report: A practical clinical definition of epilepsy. *Epilepsia*, *55*(4), 475–482. https://doi.org/10.1111/epi.12550
Kwan, P., Arzimanoglou, A., Berg, A. T., et al. (2010). Definition of drug resistant epilepsy: Consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. *Epilepsia*, *51*(6), 1069–1077. https://doi.org/10.1111/j.1528-1167.2009.02397.x
Glauser, T., Ben-Menachem, E., Bourgeois, B., et al. (2013). Updated ILAE evidence review of antiepileptic drug efficacy and effectiveness as initial monotherapy for epileptic seizures and syndromes. Epilepsia, 54(3), 551–563. https://doi.org/10.1111/epi.12074
Perucca, E., & Tomson, T. (2011). The pharmacological treatment of epilepsy in adults. The Lancet Neurology, 10(5), 446–456. https://doi.org/10.1016/S1474-4422(11)70047-3
[D6] Malaník, M., Čulenová, M., Sychrová, A., et al. (2023). Treating epilepsy with natural products: Nonsense or possibility? Pharmaceuticals, 16(8), 1061. https://doi.org/10.3390/ph16081061
Devinsky, O., Cross, J. H., Laux, L., et al. (2017). Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. *New England Journal of Medicine*, *376*(21), 2011–2020. https://doi.org/10.1056/NEJMoa1611618
[D8] Rogawski, M. A., & Löscher, W. (2004). The neurobiology of antiepileptic drugs. Nature Reviews Neuroscience, 5(7), 553–564. https://doi.org/10.1038/NRN1430
Catterall, W. A. (2014). Sodium channel mutations and epilepsy. In J. L. Noebels et al. (Eds.), Jasper’s Basic Mechanisms of the Epilepsies (4th ed.). National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/books/NBK98162/
[D10] Treiman, D. M. (2001). GABAergic mechanisms in epilepsy. Epilepsia, 42(Suppl 3), 8–12. https://doi.org/10.1046/j.1528-1157.2001.042suppl.3008.x
[D11] Meldrum, B. S. (2000). Glutamate as a neurotransmitter in the brain: Review of physiology and pathology. Journal of Nutrition, 130(4), 1007S–1015S. https://doi.org/10.1093/jn/130.4.1007S
Dravet, C. (2011). The core Dravet syndrome phenotype. Epilepsia, 52(Suppl 2), 3–9. https://doi.org/10.1111/j.1528-1167.2011.02994.x
Glauser, T. A. (2004). Ethosuximide, valproic acid, and lamotrigine in childhood absence epilepsy. New England Journal of Medicine, 362(9), 790–799. https://doi.org/10.1056/NEJMoa0902014
Vezzani, A., Balosso, S., & Ravizza, T. (2008). The role of cytokines in the pathophysiology of epilepsy. Brain, Behavior, and Immunity, 22(6), 797–803. https://doi.org/10.1016/j.bbi.2008.03.009
[D15] Waldbaum, S., & Patel, M. (2010). Mitochondrial dysfunction and oxidative stress: A contributing link to acquired epilepsy? Journal of Bioenergetics and Biomembranes, 42(6), 449–455. https://doi.org/10.1007/s10863-010-9320-9
[D16] Löscher, W., Klitgaard, H., Twyman, R. E., & Schmidt, D. (2013). New avenues for anti-epileptic drug discovery and development. Nature Reviews Drug Discovery, 12(10), 757–776. https://doi.org/10.1038/nrd4126
[D17] Marson, A. G., Al-Kharusi, A. M., Alwaidh, M., et al. (2007). The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy. The Lancet, 369(9566), 1000–1015. https://doi.org/10.1016/S0140-6736(07)60460-7
[D18] Bialer, M., & White, H. S. (2010). Key factors in the discovery and development of new antiepileptic drugs. Nature Reviews Drug Discovery, 9(1), 68–82. https://doi.org/10.1038/nrd2997
Macdonald, R. L., & Kelly, K. M. (1994). Mechanisms of action of currently prescribed and newly developed antiepileptic drugs. Epilepsia, 35(Suppl 4), S41–S50. https://doi.org/10.1111/J.1528-1157.1994.TB05955.X
Stockis, A., Lu, S., & Tonner, F. (2009). Clinical pharmacology of levetiracetam for the treatment of epilepsy. Expert Review of Clinical Pharmacology, 2(4), 365–379. https://doi.org/10.1586/ECP.09.16
[D21] Lyseng-Williamson, K. A., & Yang, L. P. H. (2007). Topiramate: A review of its use in the treatment of epilepsy. Drugs, 67(15), 2231–2256. https://doi.org/10.2165/00003495-200767150-00008
[D22] Olsen, R. W. (1981). GABA-benzodiazepine-barbiturate receptor interactions. Journal of Neurochemistry, 37(1), 1–13. https://doi.org/10.1111/j.1471-4159.1981.tb05284.x
[D23] Elterman, R. D., Shields, W. D., Mansfield, K. A., & Nakagawa, J. (2001). Randomized trial of vigabatrin in patients with infantile spasms. Neurology, 57(8), 1416–1421. https://doi.org/10.1212/WNL.57.8.1416
[D24] Steinhoff, B. J., Hamer, H., Trinka, E., et al. (2014). A multicenter survey of clinical experiences with perampanel in real life in Germany and Austria. Epilepsy Research, 108(5), 986–988. https://doi.org/10.1016/j.eplepsyres.2014.02.009
Ryvlin, P., Werhahn, K. J., Blaszczyk, B., et al. (2014). Adjunctive brivaracetam in adults with uncontrolled focal epilepsy. *Epilepsia*, *55*(1), 47–56. https://doi.org/10.1111/epi.12433
Tisserand, R., & Young, R. (2014). Essential oil safety: A guide for health care professionals (2nd ed.). Churchill Livingstone. https://doi.org/10.1016/C2010-0-67668-8
[D27] Herz, R. S. (2009). Aromatherapy facts and fictions: A scientific analysis of olfactory effects on mood, physiology and behavior. International Journal of Neuroscience, 119(2), 263–290. https://doi.org/10.1080/00207450802333953
Koutroumanidou, E., Kimbaris, A., Kortsaris, A., et al. (2013). Increased seizure latency and decreased severity of pentylenetetrazol-induced seizures in mice after essential oil administration. Epilepsy Research and Treatment, 2013, 532657. https://doi.org/10.1155/2013/532657
Malaník, M., Čulenová, M., Sychrová, A., et al. (2023). Natural products in epilepsy treatment. Pharmaceuticals, 16(8), 1061. https://doi.org/10.3390/ph16081061
[D30] Linck, V. M., da Silva, A. L., Figueiró, M., et al. (2009). Inhaled linalool-induced sedation in mice. Phytomedicine, 16(4), 303–307. https://doi.org/10.1016/j.phymed.2008.08.001
Sangtam, T. C. (2024). A review on molecular mechanism of antiepileptic drugs. International Journal for Multidisciplinary Research, 6(6). https://doi.org/10.36948/ijfmr.2024.v06i06.33763
[D32] Betts, T., & Betts, H. (1998). Seizure treatment with olfactory training: A preliminary trial. Neurological Sciences, 43(3), 285–290. https://doi.org/10.1007/s10072-022-06376-2
Koutroumanidou, E., Kimbaris, A., Kortsaris, A., et al. (2013). Increased seizure latency and decreased severity of PTZ-induced seizures in mice after essential oil administration. Epilepsy Research and Treatment, 2013, 532657. https://doi.org/10.1155/2013/532657
Kasper, S., Gastpar, M., Müller, W. E., et al. (2010). Silexan, an orally administered Lavandula oil preparation, is effective in the treatment of mixed anxiety and depression disorder. *Phytomedicine*, *17*(2), 94–99. https://doi.org/10.1016/j.phymed.2009.10.007
Devinsky, O., Cilio, M. R., Cross, H., et al. (2014). Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. *Epilepsia*, *55*(6), 791–802. https://doi.org/10.1111/epi.12631
Thiele, E. A., Marsh, E. D., French, J. A., et al. (2018). Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome. *The Lancet*, *391*(10125), 1085–1096. https://doi.org/10.1016/S0140-6736(18)30136-3
Hausman-Kedem, M., Menascu, S., & Kramer, U. (2018). Efficacy of CBD-enriched medical cannabis for treatment of refractory epilepsy in children and adolescents. Brain and Development, 40(7), 544–551. https://doi.org/10.1016/j.braindev.2018.03.013
[D38] Cavanagh, H. M. A., & Wilkinson, J. M. (2002). Biological activities of lavender essential oil. Phytotherapy Research, 16(4), 301–308. https://doi.org/10.1002/ptr.1103
[D39] Porres-Martínez, M., González-Burgos, E., Carretero, M. E., & Gómez-Serranillos, M. P. (2016). Neuroprotective properties of major terpene components of essential oils. Current Medicinal Chemistry, 23(37), 4164–4176. https://doi.org/10.2174/0929867323666160927143923
Liapi, C., Anifandis, G., Chinou, I., et al. (2007). Antinociceptive properties of 1,8-cineole and β-pinene, from the essential oil of Eucalyptus camaldulensis leaves, in rodents. Planta Medica, 73(12), 1247–1254. https://doi.org/10.1055/s-2007-990224
Baser, K. H. C. (2008). Biological and pharmacological activities of carvacrol and carvacrol bearing essential oils. Current Pharmaceutical Design, 14(29), 3106–3119. https://doi.org/10.2174/138161208786404227
[D42] Guimarães, A. G., Quintans, J. S. S., & Quintans-Júnior, L. J. (2013). Monoterpenes with analgesic activity – A systematic review. Phytotherapy Research, 27(1), 1–15. https://doi.org/10.1002/ptr.4686
Bhatt, D. L., Bhatt, M. R., & Bhatt, R. R. (2013). Thymol as an anticonvulsant modulator of GABA-A receptors. Phytomedicine, 20(8–9), 1–7. https://doi.org/10.1016/j.phymed.2013.01.001
Ali, B., Al-Wabel, N. A., Shams, S., et al. (2015). Essential oils used in aromatherapy: A systematic review. *Asian Pacific Journal of Tropical Biomedicine*, *5*(8), 601–611. https://doi.org/10.1016/j.apjtb.2015.05.007
Serafino, A., Sinibaldi Vallebona, P., Andreola, F., et al. (2008). Stimulatory effect of eucalyptus essential oil on innate cell-mediated immune response. BMC Immunology, 9, 17. https://doi.org/10.1186/1471-2172-9-17
[D46] Al-Yasiry, A. R. M., & Kiczorowska, B. (2016). Frankincense – Therapeutic properties. Advances in Hygiene and Experimental Medicine, 70, 380–391. https://doi.org/10.5604/17322693.1200553
[D47] Kunnumakkara, A. B., Sailo, B. L., Banik, K., et al. (2018). Chronic diseases, inflammation, and spices: How are they linked? Journal of Translational Medicine, 16(1), 14. https://doi.org/10.1186/s12967-018-1381-2
Srivastava, J. K., Shankar, E., & Gupta, S. (2010). Chamomile: A herbal medicine of the past with bright future. *Molecular Medicine Reports, 3*(6), 895–901. https://doi.org/10.3892/mmr.2010.377
Viola, H., Wasowski, C., Levi de Stein, M., et al. (1995). Apigenin, a component of Matricaria recutita flowers, is a central benzodiazepine receptors-ligand with anxiolytic effects. Planta Medica, 61(3), 213–216. https://doi.org/10.1055/s-2006-958058
[D50] Synergistic anticonvulsant activity of oregano, chamomile, and lavender via intranasal administration. (2025). Pharmacological Research, 2025. https://doi.org/10.1016/j.pharma.2025.05.004
Francomano, F., Caruso, A., Barbarossa, A., et al. (2019). β-caryophyllene: A sesquiterpene with countless biological properties. Applied Sciences, 9(24), 5420. https://doi.org/10.3390/APP9245420
[D52] Bahi, A., Al Mansouri, S., Al Memari, E., et al. (2014). β-Caryophyllene, a CB2 receptor agonist produces multiple behavioral changes relevant to anxiety and depression in mice. Physiology & Behavior, 135, 119–124. https://doi.org/10.1016/J.BBR.2019.112439
Kennedy, D. O., Wake, G., Savelev, S., et al. (2003). Modulation of mood and cognitive performance following acute administration of single doses of Melissa officinalis. Neuropsychopharmacology, 28(10), 1871–1881. https://doi.org/10.1038/sj.npp.1300230
Hosseinzadeh, H., & Parvardeh, S. (2004). Anticonvulsant effects of thymoquinone, the major constituent of Nigella sativa seeds, in mice. Phytomedicine, 11(1), 56–64. https://doi.org/10.1078/0944-7113-00376
McPartland, J. M., Duncan, M., Di Marzo, V., & Pertwee, R. G. (2015). Are cannabidiol and Δ9-tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. British Journal of Pharmacology, 172(3), 737–753. https://doi.org/10.1111/bph.12944
Glossary
1,8-Cineole
Monoterpene oxide in Eukalyptus/Rosemary; NO system + Nrf2; anticonvulsant, neuroprotective
AED
Antiepileptic Drug
AMPA
Alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor – ionotropic glutamate receptor; target of topiramate, perampanel
Seizure threshold
Minimal stimulus intensity that triggers an epileptic seizure; modulable by AEDs and terpenes
Apigenin
Flavonoids in chamomile; GABA-A (BZD-Site, Ki = 4 µM); anxiolytic, anticonvulsant
β-Caryophyllene
Bicyclic sesquiterpene; CB2 agonist + NF-κB inhibitor; anti-neuroinflammatory
Borneol
Bicyclic monoterpene alcohol in rosemary/camphor; GABA-A + Nav; neuroprotective
Carbamazepine
Nav Channel Blocker; First-line therapy focal epilepsy; Agranulocytosis risk
Carvacrol
Monoterpenols in Oregano/Thyme; Nav + TRPV1 + GABA-A; anticonvulsant
Cavity channel
Voltage-dependent calcium channel; T-type (Cav3.1/3.2) target of ethosuximide and valproate
CB2
Cannabinoid Receptor Type 2 – anti-neuroinflammatory; target of β-Caryophyllene and CBD
CBD
Cannabidiol – non-psychoactive phytocannabinoid; FDA/EMA-approved for Dravet and LGS
Dravet Syndrome
Severe epilepsy with SCN1A mutation; clinically approved CBD therapy (Epidiolex)
Entourage effect
Synergistic effect of multiple cannabinoids and terpenes in full-spectrum cannabis extract
Epidiolex
Pharmaceutical CBD preparation; approved for Dravet syndrome and Lennox-Gastaut syndrome
Epilepsy
Chronic neurological disease with a persistent predisposition to epileptic seizures; affects ~50 million people worldwide
Ethosuximide
T-type Ca2+ blockers; first-line therapy absence epilepsy
Fennel
Ingredient: Fennel oil; proconvulsant in high doses - hazardous substance
GABA-A
Ionotropic GABA receptor with chloride channel; target of phenobarbital, benzodiazepines, linalool, borneol
GABA-T
GABA transaminase – enzyme for GABA degradation; inhibited by vigabatrin and rosmarinic acid
GFAP
Glial Fibrillary Acidic Protein – Marker of Astrocyte Activation and Neuroinflammation
GPR55
G-Protein-Coupled Receptor 55 – Lysophosphatidylinositol Receptor; Target of CBD
Hippocampal sclerosis
Most common cause of pharmacoresistant temporal lobe epilepsy; due to repetitive seizures
Kindling
Experimental epilepsy model by repeated subthreshold stimulation
Potassium channel
Voltage-gated potassium channel; regulates repolarization and seizure threshold
Lacosamide
Nav slow inactivation; newer generation; good tolerability
Lamotrigine
Nav channel blocker + glutamate inhibition; well tolerated; first-line therapy
Levetiracetam
SV2A binder; excellent pharmacokinetics; behavioral changes as adverse events
LGS
Lennox-Gastaut Syndrome – severe epilepsy with multiple seizure types; CBD-approved
Linalool
Monoterpenes in lavender/coriander; GABA-A modulation + Nav inhibition; anticonvulsant
Linalyl acetate
Dormant in lavender; sedative, anxiolytic; synergistic with linalool
MDA
Malondialdehyde – Marker for Lipid Peroxidation and Oxidative Stress
Navigation channel
Voltage-gated sodium channel (Nav1.1-Nav1.6); main target of carbamazepine, lamotrigine, phenytoin
NF-κB
Nuclear Factor kappa B – Master Regulator of Neuroinflammation; Inhibited by BCP, 1,8-Cineole
NMDA
N-methyl-D-aspartate receptor – ionotropic glutamate receptor; target of ketamine, memantine, trans-anethole
Nrf2
Nuclear factor erythroid 2-related factor 2 – antioxidant transcription factor; activated by terpenes
P-glycoprotein
MDR1/ABCB1 – Efflux transporter; overexpressed in multidrug-resistant epilepsy
Perampanel
AMPA antagonist; effective in pharmacoresistant generalized epilepsy
Drug resistance
Failure of ≥ 2 appropriately administered AEDs; affects ~30% of epilepsy patients
Phenobarbital
GABA-A potentiation; oldest AED; inexpensive; sedation
PTZ
Pentylentetrazol – convulsant agent; standard model for testing anticonvulsant substances
Pulegone
Component Peppermint oil; hepatotoxic and proconvulsive in high doses
Rosmarinic acid
Polyphenols in Lemon Balm; GABA-T Inhibitor; antioxidant, anticonvulsant
SOD
Superoxide dismutase – antioxidant enzyme; increased by neuroprotective terpenes
Status epilepticus
Life-threatening seizure > 5 minutes or serial seizures without recovery
SV2A
Synaptic Vesicle Protein 2A – target of levetiracetam and brivaracetam; regulates neurotransmitter exocytosis
Thymol
Monoterpene phenol in thyme; GABA-A (barbiturate site) + Nav; anticonvulsant
Thymoquinone
Active ingredient black cumin seed oil; NMDA inhibition + GABA-A; anticonvulsant
Topiramate
Multimodal AEDs (Nav + GABA-A + AMPA + CA); cognitive side effects
trans-Anethole
Active ingredient: Fennel oil; GABA-A + NMDA inhibition; anticonvulsant
TRPM8
Transient Receptor Potential Melastatin 8 – Cold receptor; activated by menthol
TRPV1
Transient Receptor Potential Vanilloid 1 – Capsaicin Receptor; modulated by CBD and Carvacrol
TRPV3
Transient Receptor Potential Vanilloid 3 – Heat Receptor; Activated by Incensol (Frankincense)
Valproate
Broad-spectrum AED; inhibits Nav, GABA-T, T-type Ca2+; teratogenic