Table of contents
Updated – February 18, 2023
This blog will no longer be continued. The Corona blog 2023 deals with background information that has become known, particularly from the pharmaceutical industry and health authority institutions at an international level.
The data update is always the top priority here - articles on various topics follow below this section. The most recent post is at the top and has a red heading.
After some updates were missed due to time constraints, below are video animations of the relevant data over time from the first recorded report of a side effect to date in a chronological order, the most recent first. (Source: EMA / EMA data analysis programming).
- Pericarditis
- Creutzfeldt-Jakob disease
- Myocarditis
- Menstrual disorder
- Guillain-Barre syndrome
- Bell's palsy
- Sleep disorder
- Hallucination
- Thrombocytopenenia
- Spontaneous abortion
- Eye pain
- Loss of consciousness
- Narcolepsy
- Lymphadenopathy
- Heart rate increased
- Headaches
- Cerebral thrombosis
- Cerebral venous sinus thrombosis
- Cerebral venous thrombosis
- Deep vein thrombosis
- Mesenteric vein thrombosis
- Portal vein thrombosis
- Superior sagittal sinus thrombosis
- Herpes zoster
- Thrombosis
- Venous thrombosis
- Venous thrombosis limb
- Tinnitus
- Sudden death
- Intermenstrual bleeding
- Menstruation delayed
- Menstruation irregular
- Urticaria
The section below will no longer be updated. The development of the EMA data will be replaced by the videos above!
Data update – EMA – VAERS – WHO – Reports of side effects
* increase from November 13th, 2021 – December 3rd, 2021 at 368.653 Messages
Total reports: EMA 1,254,029 (+ 90,673*) / WHO 2,706,410 (+206,529*) / CDC/FDA 951857 (+71,451*)
Symptom: Bell's Palsy (Facial Paralysis) - Registered Reports
Increase from December 30th, 2021 - January 15th, 2022 / December 17th, 2021 - January 7th, 2022 6.620 cases
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of December 10, 2021
Source: vaers.hhs.gov (CDC / FDA) – as of December 3rd, 2021
EMA
6.113 (+ 1.350*)
WHO
7.875 (+ 3.650*)
VAERS
5.405 (+ 1.620*)
Symptom: Menstrual disorders – Registered reports
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of November 13, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of November 5th, 2021
EMA
9.849
WHO
15.660
VAERS
4.002
Symptom: Spontaneous Abortion – Registered Reports
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of November 13, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of November 5th, 2021
EMA
1.823
WHO
3.424
VAERS
2.188
Symptom: Lymphadenopathy – Registered Reports
* increase from December 18th, 2021 - December 24th, 2021 / December 10th, 2021 - December 17th, 2021 at 713 cases
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of December 24, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of December 17, 2021
EMA
50.222
WHO
94.166
VAERS
31.040
Symptom: Deaths – Registered Reports
Current data on Excess mortality in all age groups can be found daily here.
The graphics are created using data from 29 participating countries: Belgium, Denmark, Estonia, Finland, France, Germany, Germany (Berlin), Germany (Hesse), Greece, Hungary, Ireland, Israel, Italy, Luxembourg, Malta, Netherlands, Norway , Portugal, Slovenia, Spain, Sweden, Switzerland, Great Britain (England), Great Britain (Northern Ireland), Great Britain (Scotland), Great Britain (Wales) and Ukraine.
Ukraine, Germany (Berlin) and Germany (Hesse) were not included in the pooled data.
(Source: Euromomo)
* increase from March 16, 2022 - March 26, 2022 / March 5, 2022 - March 18, 2022 at 2.595 Deaths
(doubling as of March 15th / March 4th)
Sources: adrreports.eu (EMA) – as of March 26, 2022 / vigiaccess.org (WHO) – As of March 26, 2022
Source: vaers.hhs.gov (CDC / FDA) – As of March 18, 2022
EMA
23.157 (+609*)
WHO
19.088 (+736*)
VAERS
25.051 (+1.250*)
Symptom: Myocarditis (inflammation of the heart muscle) – Registered reports
* increase from November 19th, 2021 - December 3rd, 2021 / November 12th, 2021 - November 29th, 2021 at 2.088 Messages
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of November 19, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of November 12, 2021
EMA
8.292 (+1.028*)
WHO
13.371 (+1.235*)
VAERS
7.879 (+693*)
Symptom: Pericarditis (inflammation of the heart sac) – Registered reports
* increase from December 10th, 2021 – December 24th, 2021 at 10.367 Messages
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of December 24, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of December 17, 2021
EMA
9.932 (+3.205*)
WHO
16.234 (+5.306*)
VAERS
9.546 (+4.033*)
Symptom: Herpes Zoster – Registered Reports
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of November 13, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of November 5th, 2021
EMA
12.876
WHO
23.682
VAERS
9971
Symptom: Thrombosis – Registered Reports
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of November 13, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of November 5th, 2021
EMA
8.110
WHO
11.254
VAERS
6.284
Symptom: Sudden Death – Registered Reports
Sources: adrreports.eu (EMA) / vigiaccess.org (WHO) – As of November 13, 2021
Source: vaers.hhs.gov (CDC / FDA) – As of November 5th, 2021
EMA
829
WHO
1.463
VAERS
729
Assessment of the suitability of the RT‐qPCR technique for
Evidence of possible infection and
Infectivity of individuals regarding SARS-CoV-2
Current report from Dr. rer. biol. hum. Ulrike Kämmerer
The report is here of the website the Doctors and Scientists for Health, Freedom and Democracy, eV or stored here downloadable.
Falsely positive PCR test results on the test stand
Researchers Leslie C. Woodcock, P. Stallinga and Igor Khmelinskii from the Portuguese University of Algarve report in their article published in The Lancet Respiratory Medicine in November 2021 Role of exosomes in false-positive covid-19 PCR tests about their research results, which are about this link are available for download.
Data update – EMA – VAERS – WHO – Reports of side effects
Researchers Leslie C. Woodcock, P. Stallinga and Igor Khmelinskii from the Portuguese University of Algarve report in their article published in The Lancet Respiratory Medicine in November 2021 Role of exosomes in false-positive covid-19 PCR tests about their research results, which are about this link are available for download.
Comparison of reported side effects of vaccines from 1,000 cases

Restriction of fundamental rights
In the Federal Law Gazette Part I 2021 No. 83 from December 11, 2021 Law to strengthen the
Vaccination prevention against Covid-19 and changes to other regulations in connection with the Covid pandemic from December 10, 2021 become the fundamental rights
- physical integrity
- the freedom of the person
- freedom of assembly
- of freedom of movement
- the inviolability of the apartment
RESTRICTED (Download PDF):

The following changes will come into force on November 25, 2021:
Article 16 – Amendment to the Twelfth Book of the Social Code “§ 142 Transitional regulation for communal lunch catering for people with disabilities due to the Covid-19 pandemic; Authorization to issue regulations.”
Article 17 – Amendment to the Federal Supply Act
Article 18 – Amendment to the Asylum Seekers Act
The following changes will come into force on January 1st, 2022:
Article 12a – Amendment to the Third Book of the Social Code
“In Section 109 Paragraph 5 Sentence 3 the statement “31. December 2021” replaced by the information “March 31, 2022”.
“§ 421c is amended as follows: “aa) In the sentence before number 1, the words “until December 31, 2021” are replaced by the words “from January 1, 2022 to March 31, 2022”. bb) In the sentence after number 2, the words “if the entitlement to short-time work benefits arose by March 31, 2021 and” are deleted.”
The following changes will come into force on January 1st, 2023:
Article 2 – Further changes to the Infection Protection Act
“The Infection Protection Act, last amended by Article 1 of this Act, is amended as follows:
- The §§ 20a and 20b are repealed.
- § 73 is changed as follows:
a) Paragraph 1a numbers 7e to 7h are repealed
b) In paragraph 2, the information “7h” is replaced by the information “7d”.
Interview compulsory corona vaccination: pros and cons – MDR
Tuesday, November 23rd, 2021 6:50 a.m. - Duration 6:50 min.
The Interview by MDR is reproduced below as a transcript and here as Audio download available. The moderator is Tim Deisinger, his discussion partners are Prof. Peter Dabrock, Professor of Theology at the University of Erlangen Nuremberg and Dr. med. Steffen Rabe, pediatrician in Munich and board member of the Doctors for individual vaccination decisions e. v. (ÄIIE).
- Moderator, Tim Deisinger:
The big taboo that a general compulsory vaccination is no longer a thing, many people are speaking out and are in favor of it. We want to take a closer look at it this morning and also want to know your opinion on it, more on that in a moment. First of all, a kind of basis for discussion, so to speak. We want to hear two points of view.
The second will then be a pediatrician. The first is now Peter Dabrock, professor of theology at the University of Erlangen Nuremberg and was chairman of the German Ethics Council until 2020. Mr. Dabrock, how do you see that? General compulsory vaccination, yes or no?
- Prof. Peter Dabrock:
So I, um, admit that when it comes to the general compulsory vaccination, my position has changed over time and that's what we notice in these questions too: there isn't a single judgment that you've made once and then just stick to it, but you have to adapt it to the circumstances. I have been promoting this for months and also hoped that people would realize that there is minimal risk for yourself and great benefit for yourself and for others.
and that you therefore get vaccinated out of self-protection, direct protection from others and solidarity. That wasn't the case and when I heard that it had become very rigid, there was a corresponding survey three weeks ago. My position has also changed and that's why I'm now leaning towards the position of saying that we need general vaccination requirements and as quickly as possible.
- Moderator, Tim Deisinger:
But understanding for those who don't want to be vaccinated or don't want to be vaccinated yet, do you still have that?
- Prof. Peter Dabrock:
So of course you think about it, especially when you feel such massive resistance to walking, and then you always hear the two arguments: it's actually proportionate and it's not
an invasion of physical integrity. The two are also connected and I would say, firstly, when it comes to physical integrity, anyone who is against compulsory vaccination must be clear that the damage to the body if one suffers from the disease or even if others who suffer the disease will be massively larger. All serious science says that there are clearly minimal residual risks, but the benefits are significantly greater.
The other thing is, uh, that you can never make physical integrity absolute when it comes to basic rights, but rather have to bring it into a practical concordance with other basic rights and if the freedom of everyone else is massively restricted because, uh, a small group ensures that, uh, that If the virus continues to spread like this, then physical integrity, which I can understand at first glance, cannot be made absolute.
- Moderator, Tim Deisinger:
The opinion of Peter Dabok, former chairwoman of the German Ethics Council. And now we want to hear Doctor Steffen Raabe, pediatrician and adolescent doctor, board spokesman for the Association of Doctors for Individual Vaccination Decisions, Mr. Rabe, so your line of thought is already indicated in the name of the association. Can you still understand Mr. Darbrock's argument?
- Dr. Steffen Rabe
No, the argument for compulsory vaccination is completely incomprehensible to me, especially with the Covid vaccines. And when I heard Mr. Darbrock and he argued with indirect third-party protection, then that is of course the crucial sticking point. Only such an argument can even justify the consideration of compulsory vaccination and it is precisely this aspect that the Covid vaccines do not cover. The Covid vaccines provide those who want to protect themselves with temporary, not bad protection against severe disease. But you have no relevant third-party protection at all.
This means that any argument for compulsory vaccination is off the table. And if he talks about a low and minimal risk with the vaccination, then that is simply wrong. As a pediatrician, I am confronted with sitting opposite young 16, 18-year-old men to whom I have to say that if they now get vaccinated with Biontech, the only vaccine recommended and approved for them at the moment, then they are at risk The incidence of myocarditis as a direct result of this vaccination is at least 1:5000. Mr. Deisinger, we don't know of any other medication. I have not known of any other vaccine for 30 years that combines such a serious disease such as myocarditis with such a dramatically high risk. This compulsory vaccination is neither legally, morally nor medically intelligent in any way; rather, as Mr. Hans-Jürgen Pape said, quite rightly, it is an expression of helplessness and headlessness.
- Moderator, Tim Deisinger:
Then let's take helplessness when we look at the situation in the intensive care units or in hospitals in general, which is presented as a further argument that there is an emergency and there is no other way out of the emergency than by requiring vaccination.
- Dr. Steffen Rabe
But Mr. Deisinger, compulsory vaccination is not an immediate measure. The legal preparation, the political implementation and the medical effectiveness, we are lying to ourselves if we see any effect in the intensive care units within two to three weeks. We have to stop thinning out intensive care units and reducing intensive care beds. Instead of forcing them out of the profession with compulsory vaccinations, we must finally show the nurses there the appreciation they need so that they stay in their profession. And politics failed completely for two years. This catastrophe is a catastrophe with an announcement, Mr. Deisinger. We knew that this autumn would be another challenge, also for the hospitals and the intensive care units, and we quickly dismantled thousands of intensive care beds. And that should now serve as an argument for an intervention in one of the central fundamental rights, and here I vehemently contradict Mr. Dabrock, the right to physical integrity, especially in a country like Germany, which has this unfortunate past, including in the medical field with these interventions we should be very, very careful and very, very careful with this thinking.
Excipients ALC-0315 and ALC-0159 “for research purposes only”
The excipients contained in Pfizer/BioNTech Comirnaty ALC-0315 [(4-Hydroxybutyl)azanediyl]di(hexane-6,1-diyl)bis(2-hexyldecanoate) (CAS 2036272-55-4) and ALC-0159 2-[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide (CAS 1849616-42-7) is according to the manufacturer ABP Biosciences intended for research use only.
Currently available studies on these excipients:
- http://www.eurannallergyimm.com/cont/journals-articles/1043/volume-potential-culprits-immediate-hypersensitivity-reactions-4579allasp1.pdf (PDFDownload) 29.04.2021
- https://www.cell.com/molecular-therapy-family/molecular-therapy/fulltext/S1525-0016(21)00064-2?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS1525001621000642%3Fshowall%3Dtrue (PDFDownload) 04.02.2021
Federal government website – deletion of the statement “Will there be a legal obligation to vaccinate – NO”
The version dated 17.11.2021 00:39:55 was still

The version dated 19.11.2021 16:44:31 is now (here available in the original):

The above links are via WayBackMachine (https://web.archive.org) secured archive pages.
What is striking is that “vaccines continue to be monitored and tested even after they have been approved…”. The “approvals” are only – conditional – approvals and must be renewed annually until final approval (see below).
Extension of the – conditional – approvals for Covid-19 vaccines
The respective documents mentioned below and made available for download can be found in the section “European Commission procedures“ via the respective link given here “Commission Implementing Decision” in the form of a ZIP file by clicking on the respective one Document icon can be downloaded in the right column.
The first symbol stands for “Decisions“ (ZIP file starts with “dec", the second for "Annexes", corresponding "anx". The ending of the file name of the unpacked files represents the language abbreviation (de – German)
In the (second) column “Procedure type“, for example, there are entries regarding “Monthly updates“ (Information updates from the manufacturer about the product, side effects, etc.), “Corrigendum“ (translation corrections), “Rectificative decision“ (decisions on marketing protection, extensions thereof), as well as “Annual renewal“ (Extension of conditional approval).
Comirnaty – BionTech/Pfizer
The European Commission in Brussels announced the document on November 3rd, 2021 C(2021) 7992 (final), dem Commission implementing decision from November 3rd, 2021, “on the annual extension of the conditional authorization of the human medicinal product “Comirnaty – Tozinameran, COVID-19 mRNA vaccine (nucleoside modified)” granted by Decision C(2020) 9598(final) and amending that decision", with: "The conditional authorization granted by Decision C(2020) 9598(final) of December 21, 2020 is extended.“
Spikevax – Moderna
The European Commission in Brussels will share the document on October 4th, 2021 C(2021) 7305 (final), dem Commission implementing decision from October 4th, 2021, “on the annual extension of the conditional authorization of the human medicinal product “Spikevax – COVID-19 mRNA vaccine (nucleoside modified)” granted by Decision C(2020) 94(final) and amending that decision", with: "The conditional authorization granted by Decision C(2021) 94(final) of January 6, 2021 is extended.“
Vaxzevira – AstraZeneca
The European Commission in Brussels announced the document on November 9th, 2021 C(2021) 8206 (final), dem Commission implementing decision from November 9th, 2021, “on the annual extension of the conditional authorization of the human medicinal product “Vaxzevira - COVID-19 mRNA vaccine (nucleoside modified)” granted by Decision C(2020) 698(final) and amending that decision", with: "The conditional authorization granted by Decision C(2021) 698(final) of January 29, 2021 is extended.“
Covid-19 Vaccine – Janssen
Currently lies with the document C(2021) 1763 (final) only the Commission implementing decision for the conditional approval of the vaccine from March 11, 2021.
In Article 4 it states: “The period of validity of the approval is one year from the date of publication of this decision.”
Definitions from the Paul Ehrlich Institute (PEI) on COVID-19 vaccines
The links to the version from August 15, 2021 and September 7, 2021 are available via WayBackMachine (https://web.archive.org) secured archive pages, while the current version from September 23, 2021 * via link to the Original page of the PEI can be read. The Red Hand letters issued by the pharmaceutical companies are also there here available for download.
All websites listed are available here as a PDF download.
08/15/2021 – “COVID-19 vaccines protect against infections with the SARS-CoV-2 virus.” (website as Download PDF)
09/07/2021 – “COVID-19 vaccines protect against severe infections with the SARS-CoV-2 virus.“ (Website as Download PDF)
September 23, 2021 * – “COVID-19 vaccines are indicated for active immunization to prevent COVID-19 disease caused by the SARS-CoV-2 virus.“ (Website as Download PDF)
Our World in Data – Covid-19, vaccinations, deaths
On the website Our World in Data The John Hopkins University provides officially collected data on various topics worldwide, including the following statistics for Germany:
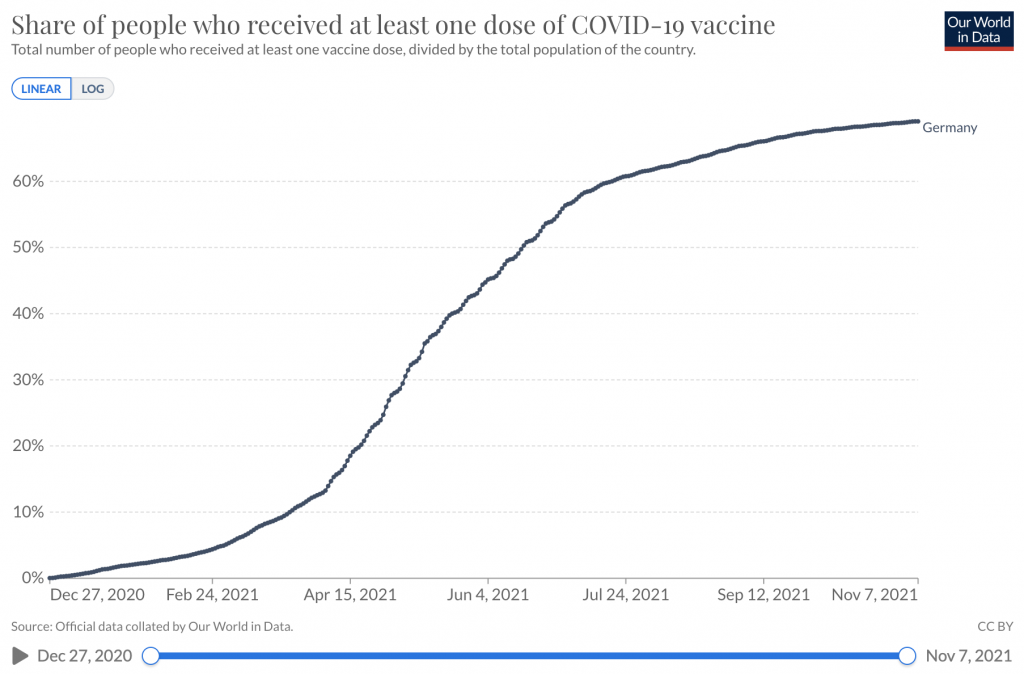
Link to statistics – Share of people who received at least one dose of COVID-19 vaccine
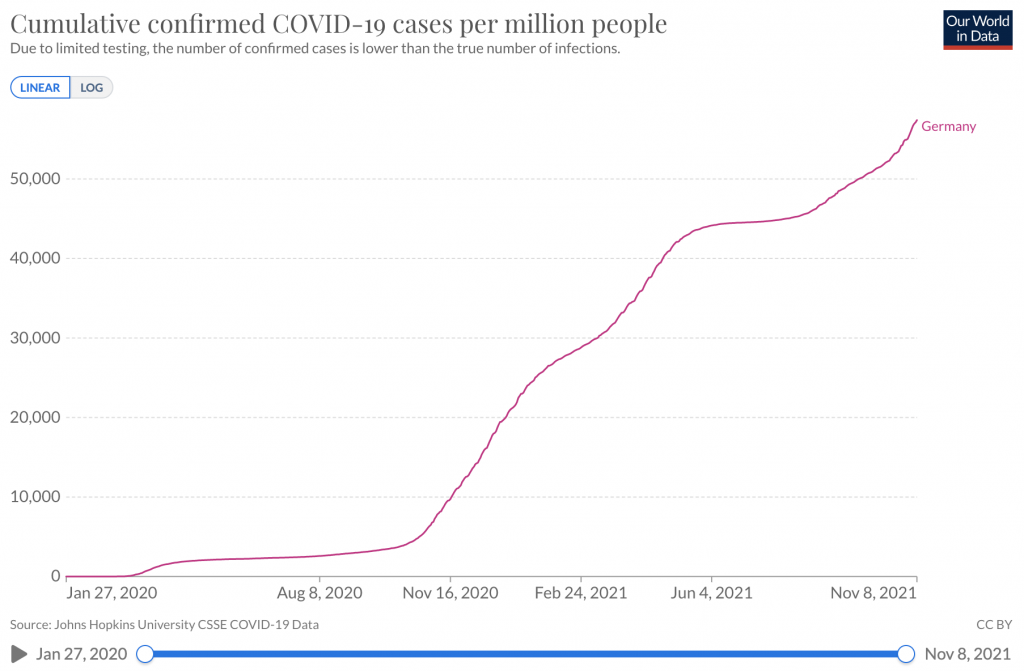
Link to statistics – Cumulative confirmed COVID-19 cases per million people
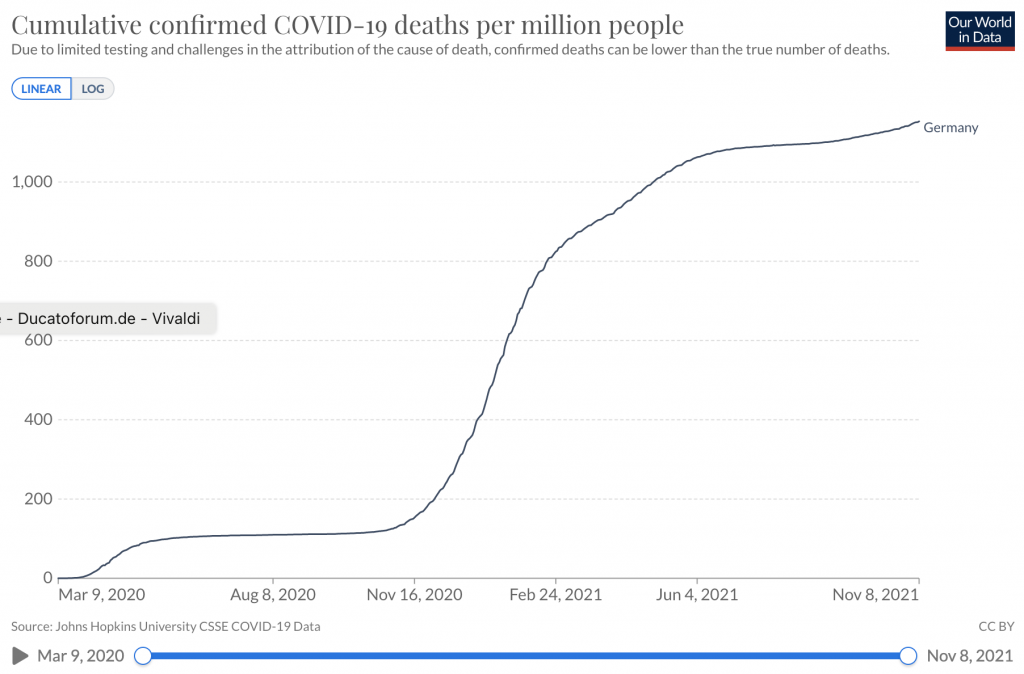
Link to statistics – Cumulative COVID-19 deaths per million people
Council of Europe – Resolution 2361/2021
The Council of Europe has in its resolution entitled Covid-19 vaccines: ethical, legal and practical considerations Recommendations have been developed that address, among other things, the fair distribution of vaccines, the voluntary nature of vaccination and non-discrimination against people who, for whatever reason, do not choose to be vaccinated.
“7.3.1. ensure that citizens are informed that vaccination is not mandatory
and that no one is put under political, social or other pressure to get vaccinated
if he doesn't want to do this himself."
“7.3.2 ensure that no one is discriminated against because they have not been vaccinated, because of possible health risks or because they do not want to be vaccinated.”
Since the Council of Europe has no legislative powers, these recommendations are not legally binding for any of the member states.
Neither a ban on compulsory vaccination nor discrimination can be derived from these recommendations - although this would be desirable in the interests of responsible citizens...
Marburg virus
Since the beginning of 2021, there have been an increasing number of articles about the Marburg virus. One appeared on February 25, 2021 publication from Elsevier Inc. in National Library of Medicine.
Almost two months later, on April 22nd, 2021, was the headline GAVI The Vaccine Alliance „The next pandemic: Marburg?“
Already in 2018 the Primer Design Ltd. a PCR test “Viral protein 35 (VP35) genes Marburgvirus genesig Standard Kit„.
Although the Marburg virus, for the first time described in 1967n was a relative of the Ebola virus, the 376 deaths at that time and only 16 since 2005 are very limited.
Against this background, the excessive push forward in the development of the vaccine to be used against the Marburg virus seems incomprehensible RiVax® through Soligenix Inc.. The rush to proceed according to the FDA's animal testing guidelines, bypassing the usual test phases 1, 2 and 3, is cause for thought.
On September 22nd, 2021, Kieran Morrissey, Dublin, Ireland, will summarize his thoughts on this topic here together.
Legal opinion on indirect compulsory vaccination
In a 111-page Legal opinion From October 4th, 2021 Prof. Dr. Dietrich Murswiek, weighing up all aspects to be taken into account, came to the summary conclusion: “The discrimination against the unvaccinated within the framework of the regulations on access to public life and within the framework of the quarantine rules violates the fundamental rights of those affected and is unconstitutional.”
Article_Li
Hospitals Should Hire, Not Fire, Nurses with Natural Immunity
BY MARTIN KULLDORFF OCTOBER 1, 2021 HISTORY, POLICY, PUBLIC HEALTH, SOCIETY 4 MINUTES
Among many surprising developments during this pandemic, the most stunning has been the questioning of naturally acquired immunity after a person has had the Covid disease.
We have understood natural immunity since at least the Athenian Plague in 430 BC. Here is Thucydides:
'Yet it was with those who had recovered from the disease that the sick and the dying found most compassion. These knew what it was from experience and had no fear for themselves; for the same man was never attacked twice—never at least fatally.' – Thucydides
We have lived with endemic coronaviruses for at least a hundred years, for which we have long-lasting natural immunity. As expected, we also have natural immunity after Covid-19 disease, as there have been exceedingly few reinfections with serious illness or death, despite a widely circulating virus.
For most viruses, natural immunity is better than vaccine-induced immunity, and that is also true for Covid. In the best study to date, the vaccinated were around 27 times more likely to have symptomatic disease than those with natural immunity, with an estimated range between 13 and 57. With no Covid deaths in either group, both natural and vaccine immunity protect well against death.
During the last decade, I have worked closely with hospital epidemiologists. While the role of doctors is to treat patients and make them well, the task of the hospital epidemiologist is to ensure that patients do not get sick while in the hospital, such as catching a deadly virus from another patient or a caretaker.
For that purpose, hospitals employ a variety of measures, from frequent hand washing to full infection control regalia when caring for an Ebola patient. Vaccinations are a key component of these control efforts. For example, two weeks before spleen surgery, patients are given the pneumococcal vaccine to minimize postoperative infections, and most clinical staff are immunized against influenza every year.
Infection control measures are especially critical for older frail hospital patients with a weakened immune system. They can become infected and die from a virus that most people would easily survive. A key rationale for immunizing nurses and doctors against influenza is to ensure that they do not infect such patients.
How can hospitals best protect their patients from Covid disease? It is an enormously important question, also relevant for nursing homes. There are some obvious standard solutions, such as separating Covid patients from other patients, minimizing staff rotation, and providing generous sick leave for staff with Covid-like symptoms.
Another goal should be to employ staff with the strongest possible immunity against Covid, as they are less likely to catch it and spread it to their patients. This means that hospitals and nursing homes should actively seek to hire staff that have natural immunity from prior Covid disease and use such staff for their most vulnerable patients.
Hence, we are now seeing a fierce competition where hospitals and nursing homes are desperately trying to hire people with natural immunity. Well, actually, not.
Instead, hospitals are firing nurses and other staff with superior natural immunity while retaining those with weaker vaccine-induced immunity. By doing so, they are betraying their patients, increasing their risk for hospital-acquired infections.
By pushing vaccine mandates, White House chief medical advisor Dr. Anthony Fauci is questioning the existence of natural immunity after Covid disease. In doing so, he is following the lead of CDC director Rochelle Walensky, who questioned natural immunity in a 2020 memorandum published by The Lancet. By establishing vaccine mandates, university hospitals are now also questioning the existence of natural immunity after Covid disease.
This is astonishing.
I work at Brigham and Women's Hospital in Boston, which has announced that all nurses, doctors and other health care providers will be fired if they do not get a Covid vaccine. Last week I spoke with one of our nurses. She worked hard caring for Covid patients, even as some of her colleagues left in fear at the beginning of the pandemic.
Unsurprisingly, she got infected, but then recovered. Now she has stronger and longer-lasting immunity than the vaccinated work-from-home hospital administrators who are firing her for not being vaccinated.
If university hospitals cannot get the medical evidence right on the basic science of immunity, how can we trust them with any other aspects of our health?
What's next? Universities questioning whether the earth is round or flat? That, at least, would do less harm.

Martin Kulldorff, Senior Scholar of Brownstone Institute, is a professor of medicine at Harvard Medical School
kulldorff@brownstone.org
Criminal complaint and criminal complaint in the BioNTech complex
To the Federal Prosecutor General at the Federal Court of Justice Dr. Peter Frank
On June 10, 2021, lawyer Tobias Ulbrich from the law firm Robert & Ulbrich, Otto Str. 12, 50859 Cologne above filed a criminal complaint and criminal complaint against “all people who developed the mRNA experimental substance from BioNTech/Pfizer known as a 'vaccine' manufactured it, distributed it, approved it for vaccination and administered it to ignorant people. In particular against:
1. Alexandra Knauer, Managing Director of the company Knauer Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin, (manufacturer of the machines for producing lipid nanoparticles)
2. Vasant Nasasimhan, CEO of Novartis AG, (patent holder for lipid nanoparticles AC – 0135 and AC 0159)
3. James Bradner, MD President of Novartis Institutes for Bio Medical Research (NIBR), developer of the lipids
4. Thomas D. Madden Ph.D. CEO Acuitas Therapeutics, manufacturer of lipids for Biontech 5. Ying K. Tam, Chief Scientific Officer Acuitas Therapeutics,
6. Sean Semple, Senior Director Pre – Clinical Research
7. Dr. Dietmar Katinger, CEO Donaustraße 99, 3400 Klosterneuburg, Austria, (manufacturer and developer of production at Biontech SE)
8. Prof. Dr. Ugur Sahin, CEO, BioNTech SE, At Goldgrube 12, 55131 Mainz
9. Sean Marett, CBO & CCO, BioNTech SE, ibid
10. Dr. Sierk Poetting, CFO & COO, BioNTech SE, ibid
11. PD Dr. Özlem Türeci, CMO, BioNTech SE, ibid
12. Ryan Richardson, CSO, BioNTech SE, ibid
13. Karin Samusch, Dermapharm AG, Lil-Dagover-Ring 7, 82031 Grünwald (Producer)
14. Hilde Neumeyer, Dermapharm AG, Lil-Dagover-Ring 7, 82031 Grünwald (Producer) 15. Dr. Hans-Georg Feldmeier, Dermapharm AG, Lil-Dagover-Ring 7, 82031 Grünwald (Producer)
16. Dr. Jürgen Ott , Dermapharm AG, Lil-Dagover-Ring 7, 82031 Grünwald (producer) 17. Mark Pfister, Production manager for Biontech at Novartis AG in Marburg (producer) 18. Dr. Sabine Brand, Siegfried Hameln, Langes Feld 13, 31789 Hameln, Germany (Producer)
19. Dr. Sven Remmerbach, Baxter Oncology GmbH, Kantstrasse 2, 33790 Halle/Westphalia (Producer)
20. Dr. Fabrizio Guidi, Chairman; Sanofi-Aventis Deutschland GmbH, Höchst Industrial Park, K703, Brüningstr. 50, 65926 Frankfurt (producer)
21. Dr. Matthias Braun, Sanofi-Aventis Deutschland GmbH
22. Oliver Coenenberg, Sanofi-Aventis Deutschland GmbH,
23. Evelyne Friday, Sanofi-Aventis Deutschland GmbH,
24. Prof. Dr. Jochen Maas, Sanofi-Aventis Deutschland GmbH,
25. Prof. Dr. Cichutek, President of the Paul Ehrlich Institute, (violation of the monitoring and warning obligation, failure to withdraw approval)
26. Prof. Dr. Vieths, Vice President of the Paul Ehrlich Institute,
27. Dr. Keller-Stanislavski, Department of Safety of Drugs and Medical Devices at the Paul Ehrlich Institute.
28. Prof. Dr. Hildt, Head of Virology Department at the Paul Ehrlich Institute
29. Prof. Dr. van Zandbergen, head of the immunology department at the Paul Ehrlich Institute
30. Dr. Add, Head of Department 3 and 4, Therapeutic Vaccines at the Paul Ehrlich Institute 31. Matthias Groote, Representative of the EMA in the European Parliament, Bergmannstraße 37, 26789 Leer,
32. Karl Broich, President of the Federal Institute for Drugs and Medical Devices and representative of the EMA in Germany, Kurt-Georg-Kiesinger-Alle 3, 53175 Bonn,
33. Ms Emer Cooke, President of the EMA, Domenico Scarlattilaaan 6, 1083 HS Amsterdam,
34. Federal Minister of Health Jens Spahn, Rochusstraße 1, 53123 Bonn,
35. Prof. Dr. Lothar H. Wieler, to be downloaded via the Robert Koch Institute,
36. Prof. Dr. Christian Drosten, to download via the Robert Koch Institute,
37. Bill and Melinda Gates,
et al
In addition, there are all non-informative vaccinators in the vaccination centers who administered the 'vaccine' without reference to the approval status and the consequences of the vaccination, which are unknown to the signatory.
Because of genocide, attempted genocide, violation of § 20 KrWKG and high treason against the federal government, among other things„
The highly interesting full text (194 pages) is here available and cites, among others, the historian Dr. Paul Schreyer, who “summarized the events of the last 20 years and presented the influence of NGOs (Non Governmental Organizations) on the preparation of the pandemic”.
LUBECAVAX – Prof. Dr. Winfried Stöcker, Lübeck
As of August 31, 2021
Winfried Stöcker was born in Upper Lusatia in 1947. Studied medicine from 1967 to 1973 in Würzburg, received his doctorate in 1976, professor at the Tongji Medical University in Wuhan since 1999, honorary professor at the University of Lübeck since 2011, founded the company EUROIMMUN Medical Laboratory Diagnostics AG 1987 with a focus on autoimmune and allergy diagnostics, as well as infection serology and molecular genetics.
Prof. Dr. Stöcker began developing an effective vaccine against SARS CoV2 at an early stage, first testing it on himself, then vaccinating his family members and finally making the self-made vaccine available to his employees.
He describes how it works on his Blog as follows (Quote):
We assume that a corona infection can be effectively prevented through a vaccination. The Lübeck vaccination uses a small, tailor-made, genetically engineered trivial antigen that the body does not have to synthesize itself, as is the case with gene ferry-based processes. It induces the formation of antibodies in the recipient's organism against exactly those structures of the virus with which it binds to the angiotensin-2 receptors of the endothelial cells in unvaccinated people. Through this blockade, the antibodies prevent the cells from becoming infected; the virus cannot find a foothold.
Vaccinations are usually carried out three times: on day zero, then after around 14 days and again after around another four weeks. The antibody concentration is measured 14 days later because we do not have the officially prescribed confidence that immune protection will have been built up by then. Over 95% of the patients ultimately show a high concentration of antibodies of the immunoglobulin class IgG against corona spike proteins, which means they are likely to be immune to corona. Immunocompromised individuals are vaccinated once or twice with a double dose - this can only be recognized by examining the serum - and half of them then achieve high titers. In addition, measurements showed that the antibodies were able to neutralize (inactivate) the corona viruses and that T-cell immunity was built up in three quarters of the cases.
He also reports on the topic of vaccine production and application by doctors in general (Quote):
In Germany, every doctor is allowed to mix an antigen with an adjuvant (only now it is a vaccine) and legally inject or apply it individually to his patient. The adiuvant holds the antigen and presents it to the immune system. Without Adiuvans, the antigen would be distributed throughout the entire organism and would therefore be diluted to the point of ineffectiveness. For functional reasons, the two components must be kept separately and freshly mixed together. However, according to the law, the doctor is not allowed to pass on the vaccine he has produced to third parties (put it on the market).
The source is listed on his blog:
medidoc GmbH
Jakob-Haringer-Strasse 1
5020 Salzburg
AUSTRIA
E-mail: info@medidoc.uk
Telephone number: +43 59333 2000
medidoc.uk
medidoc.us
medidoc.gmbh
UID: ATU33905904
Salzburg City Tax Office 114/8583
Commercial register: 45971F
Commercial register court: Salzburg Regional Court
The problem that this vaccine is not yet recognized by the EU (compared to the emergency approvals of mRNA and vector preparations) is compensated for by the fact that a T-cell immunity results, similar to that of those who have recovered.
T-cell immunity is determined and certified by appropriately equipped laboratories. This certificate serves as legally binding proof of immunity.
As long as those who have recovered are and remain on an equal footing with those who have been vaccinated by the government with regard to various restrictions/relaxations, this vaccine is a - now well-tested - alternative, without comparable side effects of the mRNA or vector preparations.
BNT162b2 vaccine: possible codon misreading, errors in protein synthesis and alternative splicing abnormalities
In a scientific one comment dated March 25, 2021, published as a preprint by AUTHOREA, downloadable as PDF in English, possible side effects of the BioNTec/Pfizer vaccine BNT162b2 are highlighted and the general effect of mRNA vaccines is presented. Here follows the German translation:
Abstract
The BNT162b2 vaccine against Covid-19 consists of an RNA of 4284 nucleotides divided into 6 sections that provide the information to create a factory of S-spike proteins used by Sars-CoV-2 (Covid- 19) used to host. Afterwards, these proteins are directed outside the cell and trigger the immune response and antibody production.
The problem is the strong alteration of the mRNA: uracil is replaced to fool the immune system with Ψ (pseudouridine); the letters of all codon triplets are replaced by a C or a G to extremely increase the speed of protein synthesis; Replacement of some amino acids with proline; Adding a sequence (3′-UTR) with unknown change.
These impairments could raise strong doubts about the presence of codon usage errors. A possible mistranslation has consequences for the pathophysiology of a variety of diseases. Furthermore, the injected mRNA is a pre-mRNA, which can give rise to multiple mature mRNAs; These are alternative splicing abnormalities that represent a direct source of serious long-term harm to human health.
In essence, what is generated may not be identical to Protein S Spike: just an error in translational decoding, misreading of codons, production of different amino acids, then proteins to cause serious long-term damage to human health, although the DNA is not modified, but in the cell nucleus and not in the cytoplasm, where the modified mRNA arrives.
However, in this case, the correlation between synthesis rate and protein expression with synthesis errors as well as the mechanism that could affect the translation of the sequence remain unclear, many studies have not yet been carried out.
introduction
Information on how the vaccine works
BioNTec/Pfizer's Sars-CoV-2 (Covid-19) vaccine called BNT162b2, but also called Tozinameran or Comirnaty, contains about 30 µg of RNA that is injected into a lipid sphere in the human body, specifically in the cytoplasm of cells , but outside the nucleus (where the DNA is located); this RNA has modified genetic information (hence modRNA), i.e. an mRNA (messenger RNA) that contains instructions to build a protein factory, clones of the protein S Spike, i.e. the protein (and only the protein, not the whole virus) of Covid-19 used to invade and infect the host. Once serially produced by the ribosomes, they are transported out of the cell beyond the lipid coating; In this way, the immune system identifies these proteins as cell invaders and attacks them by producing antibodies. It is therefore not conceivable that the vaccine induces Covid-19 or changes human DNA.
Notes on protein synthesis
Translation is generally divided into three phases: beginning, extension and end.
- The ribosome binds to the mRNA at the start codon;
- The polypeptide chain lengthens in one direction of ribosome movement by successive addition of amino acids;
- When a stop codon is found, the polypeptide is released and the ribosome dissociates.
Errors in sequence assembly and translation
Conversion of the mRNA sequence into a polypeptide depends on transfer RNA (tRNA) to transport amino acids to the ribosome. On ribosomes, tRNA pairs with mRNA through complementary base pairing between mRNA codon nucleotides and tRNA anticodon nucleotides. Once the correct tRNA is bound by a codon, it transfers its amino acid to the end of a growing polypeptide chain.
Decoding of mRNA codons by transfer RNAs (tRNAs) in the ribosome involves Watson-Crick base pairing.
The general error rates of genomic replication (approximately 10-8) are estimated to be approximately 10,000-fold lower than those of protein synthesis (approximately 10-4), and therefore in most cases mRNA translation is the key process leading to Inaccuracy of the cellular proteome. The discrepancy between error rates in DNA replication and mRNA translation may be due in part to the fact that DNA replication occurs at the level of single nucleotides (with 41 = 4 possible permutations), while the translation machinery interprets mRNA codons in triplets (with 43 = 64 possible permutations).(1)
The efficiency of the mRNA decoding machinery is also essentially regulated by the codon usage bias, which is characterized by over- or under-represented synonymous codons. Accordingly, optimizing tRNA wobble and codon usage in mRNA can significantly improve translation efficiency and accuracy.(1)
Pre- or post-mRNA translation may indirectly introduce errors in protein synthesis during transcription and post-translational processing. However, the translation machinery can directly contribute to mistranslation by tRNA misdecoding (leading to misincorporation or stop codon readthrough), tRNA misacylation (leading to incorrect tRNA-amino acid coupling), codon reassignment, or provoked by ribosomal translocation Frameshifts.(1)
Examination method
Genetic sequence analysis
The vaccine consists of 4284 nucleotides divided into 6 sections: cap is the beginning of the sequence that begins with the two GA nucleotides, which incorrectly indicates that the mRNA comes from the human cell and is therefore accepted; 5′ indicates the direction to follow for translation, while UTR indicates the area where the ribosome must rest to make proteins. In this section, the U of uracil was replaced by a molecule 1-methyl-3′-pseudouridine, marked with the sign Ψ, to bypass the immune system and prevent the degradation of the just-invaded mRNA; However, this is a factor that can lead to errors in protein production. Several Ψ-synthases are involved in the modification of specific positions, and defects in several of them have been linked to human disease (2).
Then there is the sig section, called the extended start sequence of the S-glycoprotein signal peptide, whose information is needed to direct the newly formed protein out of the cell via the endoplasmic reticulum; here too, changes are made to the triplets of nucleotides so that the RNA is accepted by the immune system, some letters that form the information, with others (usually in third place, "wiggle"), apparently "harmless synonyms" (mainly by increasing the Number of letters C and G, which encode the speed of protein synthesis). Although they specify identical amino acids, the two synonyms are not exactly the same, at least as far as translation is concerned. Mechanistic studies show that there are subtle but significant differences in how each interacts with its corresponding transfer RNA (tRNA), differences that affect both the speed and accuracy of translation.3 While it is true that 3 letters a codon and more than one codon encodes the same amino acid, it is also true that a disproportionate increase in the rate of protein production could pose a risk of serious translation errors.
Also the characters that make up the sequence related to the construction of the real spike protein S protein_mut were changed with more C and G that could be added, respecting the synonyms in the standard table of the genetic code, with substitution of amino acids Lysine (AAA) and valine (GUU) with proline (CUU) to prevent the engineered protein from collapsing. At the end of this sequence there are 2 stop codons. It is not fully proven that with this replacement the same elements are formed and misinterpretations do not occur.
3′-UTR (Untranslated Region 3 First): It was supposed to indicate the translation direction of the sequence and improve protein synthesis, however many of its functions remain unknown; Therefore, it is impossible to verify its security. What is known is stated by the WHO and is the following sentence: The 3′ UTR for the BioNTech/Pfizer vaccine was taken from “the amino-terminal enhancer of split (AES) mRNA and the mitochondrial encoded 12S ribosomal RNA”.
poly(A): We then reach the end of the sequence and encounter 30 A's, then a 10 nucleotide GCAUAUGACU linkage, followed by another 70 A's, as each mRNA can be reused multiple times by the organism.
When the A is used up, the mRNA is degraded.
All of these are proprietary modifications to increase protein expression, of which nothing is known about the actual translation performed by the organism.
Anomalies and other errors in alternative splicing
Another related problem is that the same pre-mRNA can give rise to different mature mRNAs and therefore slightly different proteins (alternative splicing abnormalities). A change in the process of protein synthesis has been found to be the cause of the development and growth of some cancers and other diseases without changing DNA in any way.
All splicing events identified in the three genes of the PHT series involve the loss of the reading frame of the messenger sequence and the introduction of a premature termination codon (PTC), which is always more than 50-55 nucleotides upstream of the last exon-exon Connection is the alternative transcripts of the NMD (nonsense-mediated mRNA decay) surveillance system. For human and rat slc15a4/PHT1, this was demonstrated by NMD inhibition experiments in various cell lines, in which the expression of alternative variants to canonical transcripts was always stabilized after inhibition.(4)
Conclusions
Possible long-term risks to human health
We can say that the sequence, in addition to being unoptimized, raises strong doubts about the presence of codon usage errors. It is possible to speculate that excessive modification aimed at extreme increase in protein expression may be the source of error in mRNA gene sequence assembly.
Alteration in tRNA availability can lead to neurodegenerative diseases (Ishimura et al., 2014), and upregulation of specific tRNAs promotes metastasis by increasing the stability of transcripts enriched in their cognate codons.(5)
Mistranslations have very serious consequences for the pathophysiology of a variety of diseases, including multiple sclerosis, neurodegeneration, mitochondrial myopathy, encephalopathy, lactic acidosis, stroke-like episodes, Parkinson's disease, and cancer (genesis, growth acceleration, and metastasis).(6)
In this case, the correlation between the 100 % increased rate of protein synthesis with the translation errors of the sequence, as well as the mechanism that influences the production of amino acids, remains obscure because many experiments have not yet been carried out.
Basically, it can be said that the code of the overall sequence is intrinsically unbalanced, too much compared to the natural viral counterpart, and too much to say that the human organism reproduces the S-spike proteins exactly, as exact identical image, which poses a risk of serious long-term damage to human health, in addition to inadequate immunization.
What is produced from this sequence is far from precisely defined, but it is written in the genes of each individual, through the ribosomal profile, how it is translated and what is produced, i.e. the benefit or harm that is caused.
References
1. Ou X, Cao J, Cheng A, Peppelenbosch MP, Pan Q (2019) Errors in translational decoding: tRNA wobbling or misincorporation? PLoS Genet 15(3):e1008017. https://doi.org/10.1371/journal.pgen.1008017
2. Biomolecules 2020, 10(5),729; https://doi.org/10.3390/biom10050729
3. Robinson R (2014) Which codon synonym is best? It may depend on what's on the menu. PLoS Biol 12(12):e1002014. doi:10.1371/journal.pbio.1002014
4. Andries, O. (2015). mRNA modification and delivery strategies to establish a platform for safe and effective gene therapy. Ghent University. Faculty of Veterinary Medicine, Merelbeke, Belgium.
5. eLife 2019;8:e45396 DOI: 10.7554/eLife.45396
6. Mafalda Santos, Patricia M. Pereira, A. Sofia Varanda, Joana Carvalho, Mafalda Azevedo, Denisa D. Mateus, Nuno Mendes, Patricia Oliveira, Fábio Trindade, Marta Teixeira Pinto, Renata Bordeira-Carriço, Fátima Carneiro, Carl Rui Vitira .ino, Olive & Manuel AS Santos (2018) Codon-misreading tRNAs promote tumor growth in mice, RNA Biology, 15:6, 773-786, DOI: 10.1080/15476286.2018.1454244
Studies confirm reprogramming of the immune system through mRNA and vector vaccines
Stephanie Seneff from the Massachusetts Institute of Technology and Greg Nigh from Naturopathic Oncology in Portland, a research team from the Helmholtz Center for Infection Research, the Hannover Medical School, the University of Bonn as well as doctors and researchers from the Erasmus Medical Center in Rotterdam come to consistent results
The tenor of the studies is the reduction of the human immune system induced by the mRNA substances with regard to the so-called tool-like receptors. These are responsible for recognizing the structures of bacterial and viral pathogens.
The study by Stephanie Seneff ea and des Research teams from the Helmholtz Institute are available for download here.
That too P.E.I (Paul Ehrlich Institute) already points out the antibodies that increase the viral load as of July 30, 2020 (!) (website as Download PDF):
“Infection-enhancing antibodies do not trigger virus elimination or neutralization, but rather enable the virus to bind to so-called Fcγ receptors, which are located, among other things, on specialized immune cells (so-called phagocytes). This in turn enables the virus to be absorbed into these cells, where the viruses can then multiply. This process can lead to an increase in viral load.”
The increased risk of thrombosis caused by the mRNA and vector substances makes matters worse. The D-dimer test can be used to estimate and detect the likelihood of an increased risk of thrombosis microscopically.
The reference value for adults is <0.5 mg/l. Values between 0.5 and 3.0 mg/l are defined as slightly elevated, values > 4.0 mg/l as severely elevated.
Study WITH
– Abstract (DE translation)
“Operation Warp Speed launched two mRNA vaccines in the United States, those from Pfizer and Moderna. Preliminary data suggested high efficacy for these two vaccines, which helped legitimize the FDA's Emergency Use Authorization (EUA).
EUA (Emergency Use Authorization) by the FDA. However, the exceptionally rapid development of these vaccines through controlled trials and the mass use of these vaccines raises numerous safety concerns. In this review, we first describe the technology underlying these vaccines in detail. Subsequently, both the components of these vaccines and the intended biological response to these vaccines, including the production of the spike protein itself, and their potential relationship to a wide range of acute and long-term pathologies, such as blood disorders, neurodegenerative diseases and autoimmune diseases. In the context of these potentially induced pathologies, we discuss the importance of amino acid sequences within the spike protein that are related to the prion protein. We also provide a brief overview of
Studies showing the potential for spike protein “shedding,” the transfer of the protein from a vaccinated person to a
unvaccinated person, which causes symptoms in the latter. Finally, we will address a frequently discussed point, namely the question of whether or not these vaccines could change the DNA of those vaccinated. While there are no studies that definitively prove this, we present a plausible scenario, supported by already established routes of transformation and transport of genetic material, whereby the injected mRNA could ultimately be incorporated into the germ cell DNA, to be transmitted across generations. We conclude with our monitoring recommendations to clarify the long-term effects of these experimental drugs and to better assess the true risk-benefit ratio of these new technologies.„
…
– Conclusion (DE translation)
„Experimental mRNA vaccines are said to have great benefits, but they also pose the risk of tragic and even catastrophic, unforeseen consequences. The mRNA vaccines against SARS-CoV-2 were introduced with great fanfare, but there are many aspects of their widespread use that raise concerns. We have addressed some, but not all, of these concerns here and would like to emphasize that these concerns are potentially serious and may not become apparent for years or even across generations. To eliminate the adverse hazards described in this paper, we recommend at least adhering to the following research findings and monitoring recommendations:
- A national survey of detailed data on adverse events associated with the mRNA vaccines, with extensive financial support and well beyond the first weeks after vaccination.
- Repeated autoantibody testing in the vaccinated population. The tested autoantibodies
could be standardized and should be based on previously documented antibodies and autoantibodies that may be triggered by the spike protein. These include autoantibodies against phospholipids, collagen, actin, thyroperoxidase (TPO), myelin basic protein, tissue transglutaminase, transglutaminase and possibly others. - Immunological profiling related to cytokine balance and associated biological effects. Tests should include at least IL-6, INF-α, D-dimers, fibrinogen, and C-reactive protein.
- Studies comparing populations vaccinated with the mRNA vaccines and those not vaccinated to confirm the expected lower infection rate and milder symptoms of the vaccinated group, while comparing the rates of recorded autoimmune diseases.
- Studies to assess whether it is possible for an unvaccinated person to acquire vaccine-specific forms of the spike proteins from a vaccinated person in close proximity.
- In vitro studies to clarify whether mRNA nanoparticles can be taken up by sperm and converted into cDNA plasmids.„
Study Helmholtz Institute
– Summary (DE translation)
„Pfizer/BioNTech's mRNA-based BNT162b2 vaccine was the first registered COVID-19 and has been shown to be effective in preventing SARS-CoV-2 infections up to 95 %.
Little is known about the broad effects of the new class of mRNA vaccines, particularly whether they have combined effects on innate and adaptive immune responses. Here, we confirmed that BNT162b2 vaccination of healthy individuals elicits effective humoral and cellular immunity against multiple SARS-CoV-2 variants. Interestingly, however, the BNT162b2 vaccine also modulated the production of inflammatory cytokines by innate immune cells after the production of inflammatory cytokines by innate immune cells, both when stimulated with specific (SARS-CoV-2) and non-specific (viral, fungal and bacterial) stimuli.
The response of innate immune cells to TLR4 and TLR7/8 ligands was lower after BNT162b2 vaccination, while fungal-induced cytokine responses were stronger. In summary, the mRNA BNT162b2 vaccine results in a complex functional reprogramming of innate immune responses, which should be taken into account in the development and use of this new class of vaccines.
…
In conclusion, our data show that the BNT162b2 vaccine has effects on both the adaptive and innate branches of immunity and that these effects are different for different SARS-CoV-2 strains.
Interestingly, the BNT162b2 vaccine also causes reprogramming of the innate immune response. Consideration: In combination with strong adaptive immune responses, this could contribute to a more balanced inflammatory response during COVID-19 infection, or it could contribute to a reduced innate immune response to the virus. The BNT162b2 vaccine clearly protects against COVID-19, but the duration of this protection is not yet known, and it would be conceivable that this knowledge could be incorporated into future generations of the vaccine to improve the reach and duration of protection. Our results need to be confirmed by conducting larger cohort studies in populations with diverse backgrounds while further studies investigate the potential interactions between BNT162b2 and other vaccines.„
Red hand letters
Red hand letters are issued by pharmaceutical companies when, among other things, previously unknown drug risks have arisen or batches of drugs are being recalled for safety reasons.
This also happened with regard to all COVID-19 pharmaceuticals (vaccines):
BionTech/Pfizer
Janssen
- 26.04.2021 – https://csiag.de/wp-content/uploads/2021/09/Rote-Hand-Janssen-26.042021.pdf
- 19.07.2021 – https://csiag.de/wp-content/uploads/2021/09/Rote-Hand-Janssen-19.07.2021.pdf
Astra Zeneca
- 24.03.2021 – https://csiag.de/wp-content/uploads/2021/09/Rote-Hand-AstraZeneca-24.03.2021.pdf
- 02.06.2021 – https://csiag.de/wp-content/uploads/2021/09/Rote-Hand-AstraZeneca-02.06.2021.pdf
- 23.06.2021 – https://csiag.de/wp-content/uploads/2021/09/Rote-Hand-AstraZeneca-23.06.2021.pdf
Medical information / leaflets for COVID vaccines
Each batch of a pack of vaccines comes with an information leaflet and medical information. These should form the basis for information before a vaccination is carried out.
The medical information from the manufacturers is made available here in the form of the EMA link and as a PDF download link. Information leaflets are currently not available online or are no longer available.
- Comirnaty – BioNTech
- Information leaflet (PDF)
- Medical information (Appendix I – EMA) – Information for users – (PDF)
- Johnson & Johnson / Janssen
- Information leaflet (PDF)
- Medical information (Appendix I – EMA) – (PDF)
- Spikevax – Moderna
- Information leaflet (PDF)
- Medical information (Appendix I – EMA) – (PDF)
- Vaxzevria – AstraZeneca
- Information leaflet (PDF)
- Medical information (Appendix I – EMA) – Medical information (Manufacturer) - (PDF)
In the information provided by the manufacturer above, the purpose of the vaccine is predominantly defined as “preventing COVID diseases”. Complete protection against future infection with COVID is not guaranteed.
AstraZeneca also points out the topic of “religious beliefs”:
“Everyone should decide for themselves whether their treatment is compatible with their own religious beliefs.”
Autopsy results of a person who died shortly after the BioNTech vaccination
Under the title First case of postmortem study in a patient vaccinated against SARS-CoV-2 was born on April 16, 2021, the result of a study carried out in cooperation with the Institute of Pathology, University Hospital OWL of the University of Bielefeld, Campus Lippe, Röntgenstr. 18, D-32756 Detmold and the Institute of Pathology, KRH Hospital Nordstadt, Hannover, Germany published the autopsy here is made available as a PDF in the original.
The full text follows in German translation:
Summary
A previously asymptomatic 86-year-old man received the first dose of the BNT162b2 mRNA COVID-19 vaccine. He died 4 weeks later of acute renal and respiratory failure. Although he had no COVID-19-specific symptoms, he tested positive for SARS-CoV-2 before his death. Spike protein (S1) antigen binding showed significant levels for immunoglobulin (Ig) G, while nucleocapsid IgG/IgM was not triggered. Acute bronchopneumonia and tubular failure were assigned as the cause of death at autopsy; However, we did not observe any distinctive morphological features of COVID-19. Postmortem molecular mapping using real-time polymerase chain reaction revealed relevant SARS-CoV-2 cycle thresholds in all organs examined (oropharynx, olfactory mucosa, trachea, lungs, heart, kidney and cerebrum) except liver and olfactory bulb. These results may indicate that the first vaccination induces immunogenicity but not sterile immunity.
We report on an 86-year-old male resident of a retirement home who was vaccinated against SARS-CoV-2. Previous medical history included systemic arterial hypertension, chronic venous insufficiency, dementia and prostate cancer. On January 9, 2021, the man received the lipid nanoparticle-formulated, nucleoside-modified RNA vaccine BNT162b2 at a dose of 30 μg. He showed no clinical symptoms that day and in the following 2 weeks (Table 1). On day 18, he was hospitalized for worsening diarrhea. Since he did not show any clinical signs of COVID-19, he was not isolated in a specific setting. Laboratory tests revealed hypochromic anemia and elevated serum creatinine levels. Antigen test and polymerase chain reaction (PCR) for SARS-CoV-2 were negative.


Gastroscopy and colonoscopy were performed to further investigate the cause of diarrhea. In particular, colonoscopy revealed an ulcerative lesion of the left colonic flexure, which was histologically diagnosed as ischemic colitis. PCR analysis on biopsy samples using a previously reported method (Kaltschmidt et al., 2021) was negative for SARS-CoV-2. Treatment was supportive with mesalazine and intravenous iron replacement. Subsequently, the patient's condition deteriorated with the development of renal insufficiency. On day 24, a patient tested positive for SARS-CoV-2 in the same hospital room as our case. On day 25, our patient tested SARS-CoV-2 positive by real-time PCR (RT-PCR), with a low cycle threshold (Ct) indicating a high viral load. Upon further analysis of the swab sample, there was no evidence of the mutated SARS-CoV-2 variants B.1.1.7, B.1.351 or B.1.1.28.1. Taken together, it appears that the patient was infected by the patient in his hospital room. Our patient now presented with fever and difficulty breathing, and pulmonary auscultation showed crackles. Despite starting supplemental oxygen (2 L per minute) and antibiotic therapy with ceftriaxone, the patient died the following day from acute renal and respiratory failure.
Assessment of immunogenicity by measuring spike protein (S1) antigen-binding mungglobulin (Ig) G in serum samples obtained on day 25 showed an antibody response (8.7 U/ml, reference value 1.0 U/ml; Roche ECLIA™). These results indicate that the patient had already developed relevant immunogenicity through vaccination.
Postmortem studies showed acute bilateral bronchopneumonia with abscesses sometimes surrounded by bacterial cocci (Figure 1). There were no findings of commonly reported manifestations of COVID-19-associated pneumonitis. In the heart we found biventricular hypertrophy (weight 580 g) and histologically diagnosed ischemic cardiomyopathy. We have detected transthyretin-type amyloidosis in the heart and, to a lesser extent, in the lungs. The kidneys showed both chronic damage with arteriolosclerosis and interstitial fibrosis and acute renal failure with hydropic tubular degeneration. Brain examination revealed left parietal pseudocystic tissue necrosis, which was diagnosed as an old infarct area.

The above figure stands here Available for download in high resolution as a PDF.
We performed molecular mapping of 9 different anatomical parts of formalin-fixed paraffin-embedded tissue as previously described (Kaltschmidt et al., 2021). RNA was extracted from paraffin sections using the Maxwell RSC (Promega, Madison, WI, USA). The multiplex RT-PCR analysis targeted 2 independent genes of the SARS-CoV-2 genome (fluorotype SARS-CoV-2 plus kit; HAIN/Bruker, Nehren, Germany): RNA-dependent RNA polymerase (target 1 ) and nucleopeptide (target 2). The negative cutoff value was Ct >45. We examined 9 different tissue samples for known and relevant routes of viral spread in the human body (Figure 1). To avoid cross-contamination, each sample was directly embedded in separate tissue cassettes and fixed separately in 4% phosphate-buffered saline-buffered formalin. We demonstrated viral RNA in almost all organs examined except the liver and olfactory bulb (Figure 1).
To the authors' knowledge, a detailed autopsy study including molecular viral mapping of a patient vaccinated against SARS-CoV-2 with a positive SARS-CoV-2 test post-vaccination has not been reported previously. We propose that a single treatment with BNT162b2b2 RNA vaccine induced significant immunogenicity, as reflected in the reported spike protein-based neutralizing IgG serum levels. From the weeks before vaccination, through vaccination (Day 1), until shortly before death (Day 24), the patient was free of clinical symptoms typically attributed to COVID-19. In addition, blood tests did not show IgM titer, which is generally observed 7-14 days after symptom onset (Kim et al., 2020). However, the patient tested positive for SARS-CoV-2. Both the ct value measured in nasopharyngeal swabs and the values measured in formalin-fixed paraffin-embedded autopsy samples indicate viral load and suggest transmissibility. Since our patient died approximately 2 days after his first positive SARS-CoV-2 test result, we assume that the molecular mapping data reflects an early stage of the viral infection. An early stage of infection could also explain why various regions such as the olfactory bulb and liver were not (yet) affected by systemic viral spread.
We have not yet observed any distinctive morphological features of COVID-19 that have been reported in large-scale morphological autopsy studies (Schaller et al., 2020, Edler et al., 2020, Ackermann et al., 2020). We did not find typical signs of diffuse alveolar damage in the lungs, but we identified extensive acute bronchopneumonia, possibly of bacterial origin. We concluded that the patient died of bronchopneumonia and acute renal failure.
Our results are consistent with previous findings from animal models that immunization against SARS-CoV-2 by vaccination appeared to reduce the severity of pathogenesis, particularly with regard to severe lung disease, while viral RNA persisted in nasal swabs (Van Doremalen et al., 2020 , Vogel et al., 2021). Recently, Amit et al. (2021) Results of a clinical trial in healthcare workers using the BNT162b2 vaccine, which showed significant early reductions in SARS-CoV-2 infection and symptomatic COVID-19 rates after administration of the first vaccine dose.
Of the main adverse reactions in patients vaccinated against SARS-CoV-2, local effects predominate and severe systemic reactions are rarely described (Yuan et al., 2020). However, recent reports of an increased risk of blood clots, particularly cerebral venous sinus thrombosis in the case of the Oxford-AstraZeneca vaccine (Mahase 2021), raised a debate about the safety of the COVID-19 vaccine in general. A comprehensive analysis of autopsy data needs to be conducted to provide more detailed insights into fatal side effects and deaths associated with vaccinations.
In conclusion, the results of our autopsy case study in a patient with mRNA vaccine confirm the view that immunogenicity can already be induced by the first vaccination against SARS-CoV-2, while sterile immunity is not sufficiently developed.
Acknowledgments
We would like to thank you for the expert technical support provided by Ralf Bode and Nadine Weber (OWL University Hospital of Bielefeld University, Lippe Campus, Detmold).
credentials
- Ackermann M., Verleden SE, Kuehnel M., Haverich A., Welte T., Laenger F. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020;383:120–128. doi: 10.1056/NEJMoa2015432. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Amit S., Regev-Yochay G., Afek A., Kreiss Y., Leshem E. Early rate reductions of SARS-CoV2-infection and COVID-19 in BNT162b2 vaccine recipients. Lancet. 2021;397(10277):875–877. doi: 10.1016/S0140-6736(21)00448-7. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Edler C., Schröder AS, Aepfelbacher M., Fitzek A., Heinemann A., Heinrich F. Dying with SARS-CoV2 infection – an autopsy study of the first consecutive 80 cases in Hamburg, Germany. Int J Legal Med. 2020;134:1275–1284. doi: 10.1007/s00414-020-02336-7. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kaltschmidt B., Fitzek ADE, Schaedler J., Förster C., Kaltschmidt C., Hansen T. Hepatic vasculopathy and regenerative responses of the liver in fatal cases of COVID-19. Clin Gastroenterol Hepatol. 2021 doi: 10.1016/j.cgh.2021.01.044. In press. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kim DS, Rowland-Jones S, Gea-Mallorqui E. Will SARS-CoV-2 infection elicit long-lasting protective or sterilizing immunity? Implications for vaccine strategies. Front Immunol. 2020;11:571481. doi: 10.3389/fimmu.2020.571481.eCollection2020. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Mahase E. Covid-19: AstraZeneca vaccine is not linked to increased risk of blood clots, finds European Medicine Agency. BMJ. 2021;372:n774. doi: 10.1136/bmj.n774. [PubMed] [CrossRef] [Google Scholar]
- Schaller T., Hirschbühl K., Burkhardt K., Braun G., Trepel M., Märkl B. Postmortem examinations of patients with COVID19. JAMA. 2020;323:2518–2520. doi: 10.1001/jama.2020.8907. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Van Doremalen N, Lambe T, Spencer A, Belij-Rammersdorfer S, Purushotham JN, Port JR ChAdOx1 nCoV-19 vaccine prevents SARS-CoV-2 pneumonia in rhesus macaques. Nature. 2020;586:578–582. doi: 10.1101/2020.05.13.093195. [PubMed] [CrossRef] [Google Scholar]
- Vogel AB, Kanevsky I, Che Y, Swanson KA, Muik A, Vormehr M. Immunogenic BNT162b vaccines protect rhesus macaques from SARS-CoV-2. Nature. 2021;592(7853):283–289. doi: 10.1101/2020.12.11.421008. [PubMed] [CrossRef] [Google Scholar]
- Yuan P., Ai P., Liu Y., Ai Z., Wang Y., Cao W. Safety, tolerability, and immunogenicity of COVID19 vaccines: a systematic review and meta-analysis. medRxiv. 2020 doi: 10.1101/2020.11.03.20224998. Preprint. [CrossRef] [Google Scholar]
Research results on dandelion extract – inhibits binding of spike proteins
Text excerpt/translation from the article “RESEARCH: Dandelion leaf extract blocks spike proteins from binding to the ACE2 cell surface receptor„:
SARS-CoV-2 spike proteins can be neutralized by a common “weed” that is banned from lawns every year. A German woman University study revealed that dandelion (Taraxacum officinale) can block the binding of spike proteins to the ACE2 cell surface receptors in human lung and kidney cells. The water-based dandelion extract, derived from the dried leaves of the plant, was effective against the spike protein D614 and a variety of mutant strains, including D614G, N501Y, K417N and E484K.
Here is the German translation of the original study (PDF - English) :
Summary:
On March 11, 2020, coronavirus disease 2019 (COVID-19), caused by the SARS-CoV-2 virus, was declared a global pandemic by the World Health Organization (WHO). To date, new “variants of concern” of SARS-CoV-2, Great Britain (B.1.1.7), the South African (B.1.351) or Brazilian (P.1) variants, are spreading rapidly. All of them contain multiple mutations in the ACE2 receptor recognition site of the spike protein, compared to the original Wuhan sequence, which is of great importance because of its immune defense potential. Here, we report the effectiveness of dandelion (Taraxacum officinale) to block the protein-protein interaction of Spike S1 with the human ACE2 cell surface receptor. This could be shown for the original spike D614, but also for its mutated forms (D614G, N501Y and mixture of K417N, E484K, N501Y) in human HEK293-hACE2 kidney and A549-hACE2-TMPRSS2 lung cells. High molecular compounds in the water-based extract are responsible for this effect. Infection of the lung cells with SARS-CoV-2 spike pseudotyped lentivirus particles was efficiently prevented by the extract, as was virus-triggered proinflammatory interleukin-6 secretion. Modern herbal monographs consider the use of this medicinal plant to be safe. Therefore, the in vitro results reported here should stimulate further research on the clinical relevance and applicability of the extract as a prevention strategy for SARS-CoV-2 infection.
SARS-CoV-2 mutates constantly during transmission between people. This could eventually lead the virus to evade existing therapeutic and prophylactic approaches that target the spike protein. We found effective inhibition of protein-protein interaction between the human viral cell entry receptor ACE2 and the SARS-CoV-2 spike protein, including five relevant mutations, by water-based extracts of common dandelion (Taraxacum officinale). This was demonstrated in vitro using human kidney (HEK293) and lung (A549) cells overexpressing the ACE2 and ACE2/TMPRSS2 proteins, respectively. The extract efficiently prevented infection of the lung cells with the pseudotyped lentivirus SARS-CoV-2. The results call for a more in-depth analysis of the effectiveness of dandelion in SARS-CoV-2 prevention and confirmatory clinical evidence.
To date, there are three rapidly spreading new variants of SARS-CoV-2, first reported in the United Kingdom (variant B.1.1.7), South Africa (variant B.1.351) and Brazil (variant P.1), all of them share the N501Y mutation in the spike protein (5). SARS-CoV-2 variants with spike protein D614G mutations now predominate worldwide. In addition to D614G, B.1.351 contains additional spike mutations, including three mutations (K417N, E484K, and N501Y) in the RBD ( 6 ). Preliminary data suggest a possible link between the observed increased mortality rate with the D614G mutation and it is suggested that a conformational change in the spike protein leads to increased infectivity ( 7 ). Free energy perturbation calculations for the interactions of the N501Y and K417N mutations, both with the ACE2 receptor and with an antibody derived from COVID-19 patients, raise important questions about the possible human immune response and the success of already available vaccines on (8). Furthermore, increased resistance of variants B.1.351 and B.1.1.7 to antibody neutralization was reported; for B.1.351, this was primarily due to the E484K mutation in the spike protein ( 9 ).
Interference with the interaction site between the spike S1 subunit and ACE2 may be an important target for therapy or prevention ( 10 ). Compounds of natural origin can provide some protection against the invasion of viral cells while having few or no side effects. Here, we report the inhibitory potential of dandelion on the binding of the spike S1 protein RBD to the hACE2 cell surface receptor and compared the effect of the original spike protein D614 with its D614G, N501Y and Mix (K417N, E484K, N501Y) -Mutations.
The common dandelion (Taraxacum officinale) belongs to the plant family Asteraceae, subfamily Cichorioideae with many varieties and small species. It is a perennial herb native to the warmer temperate zones of the Northern Hemisphere, inhabiting fields, roadsides and ruderal sites. T. officinale is consumed as a plant food, but is also used in European phytotherapy for diseases of the liver, gallbladder, digestive tract or rheumatic diseases. Modern herbal monographs consider the plant use to be safe and have evaluated the empirical use of T. officinale with a positive result. Areas of application of T. officinale are listed in the monographs of the German Commission E, the European Scientific Cooperative for Phytotherapy (ESCOP) (11, 12) and the British Herbal Medicine Association (13). The plant contains a wide range of phytochemicals, including terpenes (sesquiterpene lactones such as taraxic acid and triterpenes), phenolic compounds (phenolic acids, flavonoids, and coumarins), and polysaccharides (14). The predominant phenolic compound was found to be chicoric acid (dicaffeoyltartaric acid). The others were mono- and dicaffeoylquinic acids, tartaric acid derivatives, flavones and flavonol glycosides. In addition to these compound classes, the roots contain high amounts of inulin (15). Dosage forms include aqueous decoctions and infusions, pumped fresh plant juice, hydroalcoholic tinctures and dragees made from dry extracts, which are used as monopreparations (16), but also integral components of medicines. Our research was conducted using water-based extracts from plant leaves. We found that leaf extracts effectively blocked the spike protein or its mutated forms of the ACE2 receptor used either before or after incubation and that high molecular weight compounds are responsible for this effect. A plant of the same strain (Cichorium intybus) could have similar effects but with lower potency. The extract efficiently prevented the infection of human lung cells A549-hACE2-TMPRSS2 with the pseudotyped lentivirus SARS-CoV-2.
Results
T. officinale inhibits spike S1 – ACE2 binding
We first examined the inhibition of the interaction between SARS-CoV-2 spike protein RBD and ACE2 using extracts from T. officinale leaves. Figure 1A shows the concentration-dependent inhibition of spike S1-ACE2 binding upon treatment with T. officinale extract (EC50=12 mg/ml). Extracts from C. intybus also showed concentration-dependent binding inhibition, but with lower potency than T. officinale (EC50 = 30 mg/ml) (Fig. 1B). We then prepared two fractions of dried T. officinale and chicory leaves and separated the extracts into a high molecular weight (>5kDa) and a low molecular weight (<5kDa) fraction. As shown in Figure 1C, the bioactive compounds were mainly present in the HMW fraction. Only low activity was observed in the LMW fraction.

.
Effect of T. officinale and chicory on Sars-CoV-2 spike – ACE 2 inhibition.
AB) Concentration-dependent effect of T. officinale (TO) and C. intybus (CI) extract. CD) Effect of fractions from TO and CI leaf extract. The extracts were freeze-dried and then molecular weight fractionation was carried out. The cut-off was set at 5 kDa (HMW > 5 kDa, LMW < 5 kDa). H+L: HMW and LMW fractions; As a reference, 50 mg of dried leaves per ml of water was used. HMW and LMW fraction amounts equivalent to dried leaves were used. Binding inhibition was assessed using the ELISA technique. Bars are means + SD. Solvent control: distilled water (ad).
Using HEK293 cells overexpressing hACE2, the potential of T. officinale and C. intybus extracts to block spike binding to cells was further investigated. As can be seen from Figure 2, the pre-incubation of cells with T. officinale for one minute. effectively blocked the cell binding of Spike by 76.67 % ± 2.9 and its HMW fraction by 62.5 ± 13.4% compared to the water control. After 3 h, the inhibition was still 50 ± 13.6 % for the extract and 35.0 ± 20 % for the HMW fraction of T. officinale. Chicory extract was less effective in this test system; Binding inhibition was observed at 37 ± 20 % after 1 minute. and 5.6 ± 9.9 %.

Inhibition of binding of the S1 spike protein to human HEK293-hACE2 cells by extract preincubation.
The cells were preincubated for the indicated times with the extract of 10 mg/ml T. officinale (TO), its HMW fraction equal to 10 mg/ml extract (HMW), and 10 mg/ml C. intybus (CI). or solvent control (ad) and then treated with HIS-tagged S1 spike protein for 1 hour without a wash step in between at 4°C. Binding inhibition was determined using flow cytometry. N=3, bars are means + SD. Top left: Cytogram of gated HEK-hACE2 cells. Middle: Overlay of representative fluorescence intensity histograms for ACE2 surface expression. Top right: Overlay of representative fluorescence intensity histograms for inhibition of spike binding by the extracts or ad; positive control: 20 µg/ml soluble hACE2. Cells were stained with anti-His-tag Alexa Fluor 647 conjugated monoclonal antibody.
Cell treatment with equal amounts of spike D614 and its variants D614G and N501Y confirmed a stronger binding affinity of D614G (∼1.5-fold) and N501Y (∼3- to 4-fold) than D614 spike protein to ACE2 surface receptor of HEK293 cells (Figure 3A). Rapid pretreatment with T. officinale (within 30 s) blocked spike binding to the ACE2 surface receptor (Fig. 3B-C). After 30 seconds, this was 58.2 ± 28.7% for D614, 88.2 ± 4.6% for D614G and 88 ± 1.3% for N501Y binding inhibition by T. officinale extract. Although spike binding inhibition was observed for C. intybus extract, this was approximately 30-70% lower compared to T. officinale, depending on the spike protein examined. When binding was examined at 37°C rather than 4°C, the results were comparable for T. officinale but even weaker for chicory extract in this cell line (Figure 3D). For T. officinale and C. intybus extracts, the inhibition of spike binding was 47.90 ± 14.72 and 13.12 ± 12.37 (D614), 68.42 ± 14.53 and 8.86 ±, respectively 15.29 (D614G), 71.66 ± 7.66 and 37.56 ± 16.14 (N501Y), respectively. We also asked whether the extracts could replace spike binding to the ACE2 surface receptor of human cells. To do this, we first incubated the cells with D614, D614G or N501Y spike protein and then with the extracts. As shown in Figure 3D , T. officinale was able to effectively remove the spike from the receptor (50% on average); Chicory was much weaker then (on average 25 %). We extended our experiments to human A549-hACE2-TMPRSS2 cells and were able to confirm the results observed in HEK293-hACE2 cells for T. officinale (Figure 3D-G). This cell line was stably transfected with both the human ACE2 and TMPRSS2 genes and, interestingly, here the C. intybus extract was more effective compared to HEK-hACE2 cells. After pretreatment of the extract, the inhibition of spike binding to the cells ranged from 73.5% ± 5.2 (D614) to 86.3% ± 3.23 (N501Y) for T. officinale extract and 56.1% ± 5.28 (D614) to 63.07% ± 14.55 (N501Y) for C. intybus extract. Already at 0.6 mg/ml, T. officinale significantly blocked binding to the D614G spike protein by approximately 40% (IC50 = 1.73 mg/ml). When cells were preincubated with the spike protein before extract treatment, the results for D614 and D614G were comparable for T. officinale extract but slightly lower for N501Y (Fig. 3C–D). A mixture of the spike mutants N501Y, K417N and E484K was also tested in this setting and here too T. officinale extract blocked binding by 82.97 % ± 6.31 (extract before incubation) and 79.7 % ± 9 .15 (extract after incubation).

Binding inhibition of Spike D614 and its mutants D614G, N501Y or mixture (N501Y, K417N and E484K) to human HEK293-hACE2 and A549-hACE2-TMPRSS2 cells by extract before or after incubation.
Overlay of fluorescence intensity histogram for A) unstained HEK cells, staining control (anti-His tag A647), and cells incubated with His tag-tagged spike D614, D614G, or N501Y for 1 hour at 4°C. B) Cells preincubated with solvent control (ad), 10 mg/ml T. officinale (TO) or 10 mg/ml C. intybus (CI) for 30-60 seconds and then with His-tagged S1. Spike D614, D614G or N501Y protein were treated for 1 h without washing in between at 4°C. DG) Effect of extract incubation on HEK or A549 cells either before or after incubation with His-tagged spike D614, D614G, N501Y or mixed protein (N501Y, K417N and E484K) at 37 °C. H) Plant extracts were incubated in saliva from 4 human donors for 30 min. at 37°C. The cells were then pretreated with 5 mg/ml extracts for 60 seconds. at 37°C before incubation with His-tagged spike D614 protein for 0.5 h at 37°C. Inhibition of spike binding to human cells was assessed using flow cytometric analysis of cells stained with anti-His-tag Alexa Fluor 647 conjugated monoclonal antibody. Bars are means + SD.
Extracts incubated in human saliva for 30 min at 37°C before cell treatment had comparable effects on spike D614G inhibition (Fig. 3H), indicating good stability of the bioactive compounds in saliva.
To see whether T. officinale extract disrupts the catalytic activity of the ACE2 receptor or affects ACE2 protein expression, we treated A549-hACE2-TMPRSS2 cells with the extract for 1–24 h before cell lysis and detection. No loss of cell viability was observed after 84 h of exposure of the cells to the extract (Fig. 4A). No impairment of enzyme activity could be detected after 1 or 24 h (4B). Spike significantly decreased ACE2 protein after 6 hours (Figure 4C, black bars), and this was also true for the extract, either alone (Figure 4C, white bars) or in combination with Spike (black bars). This effect was eliminated after 24 hours (4D).

Effect of T. officinale extract on ACE2 enzyme activity and protein expression.
A) The viability of A549-hACE2-TMPRSS2 cells was determined using trypan blue cell staining after 84 h of exposure to the extract. B) Cells were incubated with TO extract or 500 ng/ml S1 protein and analyzed for enzyme activity using a fluorescence kit. CD) Cells were exposed for 6 h or 24 h to extract 500 ng/ml S1 protein without (white bars) or with (black bars) and analyzed for ACE2 protein expression using a human ACE2 ELISA kit; a. d.: Solvent control. Bars are means + SD, N ≥ 3 independent experiments.
Using a SARS-CoV-2 spike pseudotype lentivirus, we then examined whether the extract could block virus entry through spike inhibition. When pretreated with the extract, virus transduction was reduced by approximately 85% at 20 mg/ml (Fig. 5A). Under the different treatment conditions, the luminescence signal generated by virus transduction was inhibited by 70 % ± 16.7 (A), 58 % ± 9.6 (B), and 53 % ± 8.1 (C) at 10 mg/mL extract. This inhibition of virus transduction by the extract was accompanied by a significant suppression of the inflammatory response triggered by the virus, as determined by reduced secretion of the pro-inflammatory cytokine IL-6 in A549-hACE2-TMPRSS2 cells (Fig. 5D).

Viral transduction inhibition of A549-hACE2-TMPRSS2 cells by T. officinale extract.
Cells were transduced with 2.5 µl SARS-CoV-2 spike pseudotyped lentivirus (Luc reporter) for 24 h A) after pretreatment with T. officinale (TO) extract for 0.5 h, B) 3 h before addition of TO or C) without extract. The medium was then changed to fresh medium and the cells were incubated together with the extract for a further 60 h. Luminescence was detected after 1 h. (-) Negative control: bald lentiviral pseudovirion; (+) positive control: firefly luciferase lentivirus. D) The analysis of proinflammatory IL-6 cytokine secretion was carried out either after 24h of virus transduction together with extract (left), after 24h + 60h after infection with extract (middle) or after 60h after infection with extract (right) using multiplexing flow cytometric analysis. Solvent control: distilled water (ad). N ≥ 3 independent experiments.
discussion
The development of effective prevention and treatment strategies for SARS-CoV-2 infection is still in its early stages. Although the first vaccines have now received marketing approval, challenges remain regarding distribution concerns or long-term effectiveness, as well as the risk of reinfection (17, 18). However, subsequent infections may be milder than the first. In addition to vaccination against COVID-19, blocking the virus's accessibility to membrane-bound ACE2 as the primary receptor for entry into the SARS-CoV-2 target cell represents an alternative strategy for preventing COVID-19. There are different approaches here (19 ), but of course each of these treatment strategies also has its fundamental and translational challenges that must be overcome for clinical benefit. Technical hurdles include off-target potential, ACE2-independent effects, stability or toxicity (19). Compounds of natural origin could be an important resource here because they are described in the long term and many of them are considered safe. While in silico docking experiments have implicated several common natural products as ACE2 inhibitors, most of them have not yet been demonstrated to inhibit spike binding to ACE2, which could be explained by a lack of complete coverage of ACE2 binding residues by the compounds (20) . However, for glycyrrhizin, nobiletin, and neohesperidin, ACE2 binding falls partially within the RBD contact region, and therefore these have been proposed to additionally block spike binding to ACE2 ( 20 ). The same applies to synthetic ACE2 inhibitors such as N-(2-aminoethyl)-1 aziridine ethanamine (NAAE) (21). In contrast, the lipoglycopeptide antibiotic dalbavancin has now been identified as both an ACE2 binder and a SARS-CoV-2 spike ACE2 inhibitor ( 22 ); SARS-CoV-2 infection was effectively inhibited by this compound in both mouse and rhesus monkey models. Blocking of the spike-ACE2 interaction with 74 % was also shown for a hydroalcoholic pomegranate peel extract, for its main components punicalagin with 64 % and ellagic acid with 36 %. Using SARS-CoV-2 spike pseudotyped lentivirus infection of human kidney 2 (HK-2) cells, virus entry was then efficiently blocked by the peel extract ( 23 ). In the present study, we demonstrated potent inhibition of the ACE2 spike S1 RBD protein by T. officinale extracts using a cell-free assay and confirmed this finding by demonstrating efficient inhibition of ACE2 cell surface binding in two human cell lines. We observed stronger binding of variants D614G and N501Y to the ACE2 surface receptor of human cells, but all variants tested were sensitive to binding inhibition by T. officinale, either before or after use. To date, several studies indicate that the D614G virus lineage is more infectious than the D614 virus ( 24 ). The presence of characteristic mutations such as N501Y from e.g. G. the so-called UK variant B.1.1.7, lead to higher infectivity than the parent strain, which could be due to a higher binding affinity between the spike protein and ACE2 (25). Therefore, our findings on T. officinale extracts may be important here, as new virus variants of potential concern will emerge as the pandemic progresses, which may also reduce the effectiveness of some vaccines or lead to increased reinfection rates. As mentioned above, a problem in developing products to prophylactic SARS-CoV-2 infection or slow systemic viral spread is selectivity for entry of viruses with low toxicity to the host. No case of T. officinale overdose has been reported for current medical indications (11, 13, 16). The recommended dosage is 4-10 g (approximately 20-30 mg per ml of hot water) up to 3 times daily (Commission E and ESCOP). Contraindications to the use of T. officinale, according to information from the European Medicines Agency (EMA), include hypersensitivity to the Asteraceae family of plants or their active ingredients, liver and biliary diseases, including bile duct obstruction, gallstones and cholangitis, or active stomach -Intestinal diseases ulcer (16). The plant is a significant source of potassium (26, 27) and therefore cautions about the possible risk of hyperkalemia. Use in children under 12 years of age, during pregnancy and breastfeeding has not been proven due to lack of or sufficient data.
While ACE2 enzyme activity was not affected by T. officinale extract in the present study, ACE2 protein was transiently downregulated in the ACE2-overexpressing lung cell line, which requires more attention in ongoing studies. ACE2 is an important zinc-dependent mono-carboxypeptidase in the renin-angiotensin signaling pathway, which is crucial for effects on the cardiovascular and immune systems. A disruption of the angiotensin II/angiotensin (1-7) balance through inhibition of ACE2 enzyme activity or protein decrease and more circulating angiotensin II in the system is e.g. G. recognized that it promotes lung damage associated with COVID-19 disease (28, 29).
The lung is thought to be the primary target of interest, but ACE2 mRNA and protein expression have been found in epithelial cells of all oral tissues, particularly in the buccal mucosa, lip, and tongue ( 30 ). These data are consistent with the observation of very high viral loads in saliva in SARS-CoV-2-infected patients (31, 32). The oral cavity, as an essential part of the upper aerodigestive tract, is therefore considered to play a key role in the transmission and pathogenicity of SARS-CoV-2. There is a high potential that preventing virus colonization at the oral and pharyngeal mucosa could be crucial to avert further infection of other organs and the outbreak of COVID-19 (33). Commercial virucidal mouthwashes, primarily povidone-iodine, have therefore been suggested to potentially reduce the viral load of SARS-CoV-2 in infected individuals (34-36), but there are no significant clinical studies to date (36) . Blocking SARS-CoV-2 virus binding to cells in the oral cavity with T. officinale extracts may only be tolerable for a consumer for limited periods of time (e.g. product application after contact with infected people or in the event of an infection). Further physiologically relevant in vitro experiments we conducted showed that only short contact times with T. officinale extract were necessary to efficiently block SARS-CoV-2 spike binding or to remove already bound spikes from the cell surface. Further evidence of relevance was provided by experiments with pseudotyped SARS-CoV-2 spike viruses. Although the use of these pseudotyped viruses does not allow assessing the contribution of virion features such as membrane or envelope proteins to cell tropism (37), they are considered a useful tool to document the relevance of ACE2 to the cell entry steps enacted by the Spike protein are mediated.
All vaccine candidates developed aim to generate antibody (and T cell) responses against the spike protein, and spike sequences from the early Wuhan strain served as a basis here ( 38 ). However, SARS-CoV-2 constantly mutates during continuous transmission between people. Viral antigenic drift is clearly demonstrated by the recent emergence of B.1.1.7, B.1.351 or B.1.1.28 (P.1). It is evolving to potentially evade our existing therapeutic and prophylactic approaches that target the viral spike. Therefore, factors such as low toxicity in humans and effective binding inhibition of five relevant spike mutations on the human ACE2 receptor, as reported here in vitro, encourage a more in-depth analysis of the effectiveness of T. officinales in SARS-CoV-2 prevention and now requires further confirmatory clinical evidence.
Materials and methodology
plant material
The study was carried out using dried leaves of T. officinale (from Achterhof, Uplengen, Germany; batch no. 37259, B370244 and P351756). At three different locations in the Freiburg i. region. Gebr. (Germany), on July 12, 2020, and tested positive in the cell-free Spike S1-ACE2 binding assay (data not shown). C. intybus was purchased from Naturideen (Germany).
Cell lines and cultures
Human embryonic kidney 293 (HEK293) cells stably expressing hACE2 were generously provided by Prof. Dr. Stefan Pöhlmann (Göttingen, Germany). Cells were maintained in high glucose Dulbecco's modified Eagle medium (DMEM) supplemented with 10 % fetal calf serum (FCS), 100 U/ml penicillin/streptomycin, and 50 μg/ml Zeocin (Life Technologies, Darmstadt, Germany). Human A549-hACE2-TMPRSS2 cells, generated from the human lung A549 cell line, were purchased from InvivoGen SAS (Toulouse Cedex 4, France) and maintained in DMEM, high glucose supplemented with 10 % heat-inactivated FCS, 100 U /ml penicillin/streptomycin, 100 µg/ml normocin, 0.5 µg/ml puromycin and 300 µg/ml hygromycin. For subculture, all cells were first rinsed with phosphate-buffered saline (PBS), then incubated with 0.25% trypsin-EDTA until detachment. All cells were cultured at 37 °C in a humidified incubator with 5 % CO2/95 % air atmosphere.
Plant extracts
Dried plant material was weighed into an amber glass vial (Carl Roth GmbH, Germany) and mixed with HPLC grade (ad) water at room temperature (RT). The extracts were then incubated for 1 h and centrifuged at 16,000 g (3 min, RT). The supernatant was filtered (0.22 μm) before use for experiments.
Analysis of SARS-COV2 spike – ACE2 interaction inhibition using ELISA and flow cytometry
For the cell-free detection of SARS-CoV-2 Spike – ACE2 interaction inhibition, a commercially available SARS-CoV-2 inhibitor screening kit (Kat#: 16605302, Fisher Scientific GmbH, Schwerte, Germany) was used. This colorimetric ELISA assay measures the binding between immobilized SARS-CoV-2 spike protein RBD and biotinylated human ACE2 protein. Colorimetric detection is performed using streptavidin-HRP followed by TMB incubation. A SARS-CoV-2 inhibitor (hACE2) served as the methodologically verified reference.
Cell surface expression of ACE2 was determined using a human ACE2 PE-conjugated antibody (Bio-Techne GmbH, Wiesbaden-Nordenstadt, Germany) and flow cytometric analysis. To analyze SARS-CoV-2 S1 Spike RBD -ACE2 binding, 2 x 105 cells (5 x 106 cells/ml) were pretreated with plant extracts at different times. Then 500 ng/ml SARS-CoV-2 Spike S1 (Trenzyme GmbH, Konstanz, Germany), Spike S1 D614G, N50Y or mixture of K417N, E484K and N501Y (Sino Biological Europe GmbH, Eschborn, Germany) -His recombinant protein was added to each Sample was added and the samples were further incubated for 30-60 min. In another setting, cells were pretreated with 500 ng/ml SARS-CoV-2 Spike-His recombinant protein for 30 minutes before incubation with the plant extract for 30-60 seconds at 4°C or 37°C. Samples were incubated in PBS buffer containing 5% FCS. Cells were then washed once with PBS buffer containing 1% FCS at 500 × g 5 min before staining with His-tag A647 mAb (Bio-Techne GmbH, Wiesbaden-Nordenstadt, Germany) for 30 min at RT. The cells were then washed twice as described above. The cells were analyzed with a FACSCalibur (BD Biosciences, Heidelberg, Germany), 10,000 events were acquired. The mean fluorescence intensity (MFI) of each sample was determined using FlowJo software (Ashland, Oregon, USA).
Human ACE2 enzyme activity and protein quantification
A549-hACE2-TMPRSS2 (2 × 105) cells were seeded in a 24-well plate in high-glucose DMEM medium containing 10 % heat-inactivated FCS at 37 °C, 5 % CO2. Cells were then treated with T. officinale extract with/without 500 ng/ml SARS-CoV-2 S1 Spike RBD protein for 1-24 hours. The cells were then washed with PBS and lysed. 25 μg of protein was used to quantify ACE2 protein (ACE2 ELISA Kit), 5 μg for ACE2 enzyme activity (ACE2 Activity Assay Kit, Abcam, Cambridge, UK) according to the manufacturer's instructions.
Infection of A549-hACE2-TMPRSS2 cells with the pseudotyped lentivirus SARS-CoV-2
SARS-CoV-2 Spike pseudotyped lentivirus particles prepared using SARS-CoV-2 Spike (Genbank Accession #QHD43416.1) as envelope glycoproteins instead of the commonly used VSV-G were purchased from BPS Bioscience (Catalog#: 7994299). , Biomol, Hamburg). These pseudovirions also contain the firefly luciferase gene, which is driven by a CMV promoter. Thus, spike-mediated cell entry can be quantified via luciferase reporter activity. The bald lentiviral pseudovirion (BPS Bioscience #79943), in which no envelope glycoprotein is expressed, was used as a negative control. Firefly Luciferase Lentivirus (Puromycin) from BPS Bioscience (Catalog#: 79692-P) was used as a positive control for transduction. These viruses constitutively express firefly luciferase under a CMV promoter. Lung cells were grown at 0.1 × 106 cells/cm2 in 96-well plates in DMEM containing 10 % heat-inactivated FCS, 100 U/ml penicillin/streptomycin, 100 µg/ml normocin, 0.5 µg/ml puromycin and 300 µg/ ml. sown hygromycin overnight. The medium was replaced with DMEM + 10 % heat-inactivated FCS and the cells were treated with ad or T. officinale extract either 30 min before or 3 h after addition of 2.5 µl of the lentivirus particles. After 24 h of virus particle incubation, the medium was removed by washing with PBS, fresh medium was added, and the cells were incubated for another 60 h with the addition of ad or T. officinale extract. Luminescence was detected within 1 h using the one-step luciferase reagent from BPS according to the manufacturer's protocol in a Tecan multiplate reader (Tecan Group Ltd, Crailsheim, Germany).
Quantification of cytokine release by multiplex bead technique
Quantification of cytokine release by multiplex bead techniqueAfter 24 h of SARS-CoV-2 spike pseudotyped lentivirus transduction and 60 h after infection of A549-hACE2-TMPRSS2 cells, supernatants were collected and stored at -80 °C until analysis Cytokine secretion with the human MACSplex. stored Cytokine 12 kit (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany) according to manufacturer's protocol.
Molecular weight fractionation from plant extracts
Dried plant leaf extracts were prepared by adding double-distilled water (5 mL) to plant material (500 mg each). The samples were incubated in the dark at room temperature (RT) for 60 min, followed by centrifugation at 16,000 g for 3 min. The supernatants were collected and membrane filtered (0.45 μm), resulting in the extracts. Aliquots were freeze-dried for 48 hours to determine their yield by weight. The extracts were then further separated into a high molecular weight (HMW) and low molecular weight (LMW) fraction using a centrifugation tube with an insert with a molecular weight cut-off filter (5 kDa, Sartorius Stedim Biotech, Göttingen, Germany). became. . Each HMW fraction was purified by rinsing with 20 mL of water, yielding the HMW fractions as well as LMW. The fractions were freeze-dried, their yield determined by weight and stored at -20°C until use.
Determination of cell viability using trypan blue staining
Determination of cell viability using trypan blue Cell viability was assessed using the trypan blue dye exclusion test as previously described (Odongo et al., 2017). Briefly, A549-hACE2-TMPRSS2 cells were cultured for 24 h and then exposed to extracts or solvent control (ad) for 84 h.
Statistical analysis
Results were analyzed using GraphPad Prism 6.0 software (La Jolla, California, USA). Data were presented as mean + SD. Statistical significance was determined by one-way ANOVA test followed by Bonferroni correction. P values < 0.05 () were considered statistically significant and <0.01 (*) considered highly statistically significant.
Author contributions
Study design and conception: EL; Experimental design, data acquisition, data analysis: HTT, EL, NPKL; Preparation of extract fractions: CD, MG; Writing the first draft of the manuscript: EL. All authors commented on previous versions of the manuscript.
Acknowledgments
The authors thank Prof. Dr. Stefan Pöhlmann (German Primate Center, Göttingen, Germany) for providing human embryonic kidney 293 (HEK293) cells that stably express hACE2.
Sources
- 1.↵Lu R, et al. (2020) Genomic characterization and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 395(10224):565–574.CrossRefPubMedGoogle Scholar
- 2.↵Paules CI, Marston HD, & Fauci AS (2020) Coronavirus Infections-More Than Just the Common Cold. JAMA 323(8):707–708.CrossRefPubMedGoogle Scholar
- 3.↵Berlin DA, Gulick RM, & Martinez FJ (2020) Severe Covid-19. N Engl J Med 383(25):2451–2460.CrossRefPubMedGoogle Scholar
- 4.↵Huang Y, Yang C, Xu XF, Xu W, & Liu SW (2020) Structural and functional properties of SARS-CoV-2 spike protein: potential antiviral drug development for COVID-19. Acta Pharmacol Sin 41(9):1141–1149.CrossRefPubMedGoogle Scholar
- 5.↵Grubaugh ND, Hodcroft EB, Fauver JR, Phelan AL, & Cevik M (2021) Public health actions to control new SARS-CoV-2 variants. Cell.Google Scholar
- 6.↵Zhou D, et al. (2021) Evidence of escape of SARS-CoV-2 variant B.1.351 from natural and vaccine-induced sera. Cell.Google Scholar
- 7.↵Becerra-Flores M & Cardozo T (2020) SARS-CoV-2 viral spike G614 mutation exhibits higher case fatality rate. International journal of clinical practice 74(8):e13525.Google Scholar
- 8.↵Fratev F (2020) The N501Y and K417N mutations in the spike protein of SARS-CoV-2 alter the interactions with both hACE2 and human derived antibody: A Free energy of perturbation study. bioRxiv:2020.2012.2023.424283.Google Scholar
- 9.↵Ho D, et al. (2021) Increased Resistance of SARS-CoV-2 Variants B.1.351 and B.1.1.7 to Antibody Neutralization. Res Sq.Google Scholar
- 10.↵Perrotta F, Matera MG, Cazzola M, & Bianco A (2020) Severe respiratory SARS-CoV2 infection: Does ACE2 receptor matter? Respir Med 168:105996.CrossRefGoogle Scholar
- 11.↵ESCOP (2003) “Taraxaci folium” and “Taraxaci radix”. Monographs on the Medicinal Uses of Plant Drugs. (Thieme, Stuttgart) second ed., Ed pp 499–504.Google Scholar
- 12.↵Blumenthal M, Busse WR, Goldberg A, Gruenwald J, Hall T, Riggins CW, Rister RS. (eds) “Dandelion herb” and “Dandelion root with herb” In: The Complete German Commission E Monographs. Therapeutic Guide to Herbal Medicines. American Botanical Council, Austin, Texas 1998; 118-120.13. Association BHM (1990) “Dandelion Leaf” and “Dandelion Root”. British Herbal Pharmacopoeia 1:37–39.Google Scholar
- 14.↵Gonzalez-Castejon M, Visioli F, & Rodriguez-Casado A (2012) Diverse biological activities of dandelion. Nutr Rev 70(9):534–547.PubMedGoogle Scholar
- 15.↵Schutz K, Carle R, & Schieber A (2006) Taraxacum–a review on its phytochemical and pharmacological profile. J Ethnopharmacol 107(3):313–323.CrossRefPubMedWeb of ScienceGoogle Scholar
- 16.↵European Medicinal Agency (EMA) CoHMPH (2009) Assessment report on Taraxacum officinale Weber ex Wigg., folium. HMPC/579634/2008.Google Scholar
- 17.↵To KK, et al. (2020) COVID-19 re-infection by a phylogenetically distinct SARS-coronavirus-2 strain confirmed by whole genome sequencing. Clin Infect Dis. Aug 25:ciaa1275. doi: 10.1093/cid/ciaa1275.CrossRefPubMedGoogle Scholar
- 18.↵Edridge AWD, et al. (2020) Coronavirus protective immunity is short-lasting. medRxiv:2020.2005.2011.20086439.Google Scholar
- 19.↵Jia H, Neptune E, & Cui H (2020) Targeting ACE2 for COVID-19 Therapy: Opportunities and Challenges. American journal of respiratory cell and molecular biology. Dec 9. doi: 10.1165/rcmb.2020-0322PS.CrossRefGoogle Scholar
- 20.↵Zhou J & Huang J (2020) Current Findings Regarding Natural Components With Potential Anti-2019-nCoV Activity. Frontiers in Cell and Developmental Biology 8:589.Google Scholar
- 21.↵Huentelman MJ, et al. (2004) Structure-based discovery of a novel angiotensin-converting enzyme 2 inhibitor. Hypertension 44(6):903–906.CrossRefGoogle Scholar
- 22.↵Wang G, et al. (2021) Dalbavancin binds ACE2 to block its interaction with SARS-CoV-2 spike protein and is effective in inhibiting SARS-CoV-2 infection in animal models. Cell Res 31(1):17–24.Google Scholar
- 23.↵Tito A, et al. (2020) A pomegranate peel extract as inhibitor of SARS-CoV-2 Spike binding to human ACE2: a promising source of novel antiviral drugs. bioRxiv:2020.2012.2001.406116.Google Scholar
- 24.↵Korber B, et al. (2020) Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell 182(4):812–827 e819.CrossRefPubMedGoogle Scholar
- 25.↵Santos JC & Passos GA (2021) The high infectivity of SARS-CoV-2 B.1.1.7 is associated with increased interaction force between Spike-ACE2 caused by the viral N501Y mutation. bioRxiv:2020.2012.2029.424708.Google Scholar
- 26.↵Hook I, McGee A, & Henman M (1993) Evaluation of Dandelion for Diuretic Activity and Variation in Potassium Content. International Journal of Pharmacognosy 31(1):29–34.Google Scholar
- 27.↵Escudero NL, De Arellano ML, Fernández S, Albarracín G, & Mucciarelli S (2003) Taraxacum officinale as a food source. Plant Foods for Human Nutrition 58(3):1–10.PubMedWeb of ScienceGoogle Scholar
- 28.↵Imai Y, et al. (2005) Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature 436(7047):112–116.CrossRefPubMedWeb of ScienceGoogle Scholar
- 29.↵Kuba K, et al. (2005) A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med 11(8):875–879.CrossRefPubMedWeb of ScienceGoogle Scholar
- 30.↵Zhong M, et al. (2020) ACE2 and Furin Expressions in Oral Epithelial Cells Possibly Facilitate COVID-19 Infection via Respiratory and Fecal-Oral Routes. Front Med (Lausanne) 7:580796.Google Scholar
- 31.↵ To KK-W, et al. (2020) Consistent Detection of 2019 Novel Coronavirus in Saliva. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 71(15):841–843.CrossRefPubMedGoogle Scholar
- 32.↵Yoon JG, et al. (2020) Clinical Significance of a High SARS-CoV-2 Viral Load in the Saliva. J Korean Med Sci 35(20):e195–e195.CrossRefGoogle Scholar
- 33.↵Wolfel R, et al. (2020) Virological assessment of hospitalized patients with COVID-2019. Nature 581(7809):465–469.CrossRefPubMedGoogle Scholar
- 34.↵Seneviratne CJ, et al. (2020) Efficacy of commercial mouth-rinses on SARS-CoV-2 viral load in saliva: randomized control trial in Singapore. Infection:1–7.Google Scholar
- 35.↵ de Toledo Telles-Araujo G, Caminha RDG, Kallas MS, Sipahi AM, & da Silva Santos PS (2020) Potential mouth rinses and nasal sprays that reduce SARS-CoV-2 viral load: What we know so far? Clinics (Sao Paulo) 75:e2328.Google Scholar
- 36. Carrouel F, et al. (2021) Antiviral Activity of Reagents in Mouth Rinses against SARS-CoV-2. Journal of dental research 100(2):124–132.Google Scholar
- 37.↵Joglekar AV & Sandoval S (2017) Pseudotyped Lentiviral Vectors: One Vector, Many Guises. Hum Gene Ther Methods 28(6):291–301.CrossRefGoogle Scholar
- 38.↵Krammer F (2020) SARS-CoV-2 vaccines in development. Nature 586(7830):516–527.CrossRefPubMedGoogle Scholar
Council of Europe – Resolution 2361 (2021) – No compulsory vaccination
Title: Covid-19 vaccines: ethical, legal and practical considerations
In this resolution you can read under 7.1.1 “ensure high-quality trials that are sound and conducted in an ethical manner in accordance with the relevant provisions of the Convention for the Protection of Human Rights and Dignity of the Human Being with regard to the Application of Biology and Medicine: Convention on Human Rights and Biomedicine (ETS No. 164, Oviedo Convention) and its Additional Protocol concerning Biomedical Research (CETS No. 195), and which progressively include children, pregnant women and nursing mothers;".
Section 7.1.1 requires that high-quality and ethical studies be ensured in accordance with the relevant provisions of the Convention for the Protection of Human Rights and the Dignity of Persons with regard to the Application of Biology and Medicine, in accordance with the Convention on Protection of human rights and biomedicine (SEV No. 164, Oviedo Convention (Link) – PDF) and the Additional Protocol on Biomedical Research (SEV No. 195 (Link) – PDF), which eventually includes children, pregnant women and breastfeeding mothers.
The Oviedo Agreement CETS 164 (SEV 1164) of April 4, 1997 calls for in Chapter IV, Article 13, - "Interventions on the human genome - An intervention seeking to modify the human genome may only be undertaken for preventive,
diagnostic or therapeutic purposes and only if its aim is not to introduce any modification in the genome of any descendants.”
It is clearly defined here that an intervention to change the human genome is only for preventative,
diagnostic or therapeutic purposes and only if it is not aimed at changing the genome of offspring.
Article 13 – Interventions on the human genome
An intervention seeking to modify the human genome may only be undertaken for preventive,
diagnostic or therapeutic purposes and only if its aim is not to introduce any modification in
the genome of any descendants.
Point 7.3.1 says “ensure that citizens are informed that the vaccination is not mandatory and that no one is under political, social or other pressure to be vaccinated if they do not wish to do so”.
In other words, the aim is to ensure that citizens are informed that vaccination is NOT compulsory and that no one will be politically, socially or otherwise pressured to get vaccinated if they do not wish to do so.
Point 7.5.1 points out that “put in place independent vaccine compensation programs to ensure compensation for undue damage and harm resulting from vaccination;” independent compensation programs must be installed from which compensation for inappropriate vaccine damage and harm resulting from vaccination is to be paid .
Point 7.5.2 states “use vaccination certificates only for their designated purpose of monitoring vaccine efficacy, potential side effects and adverse events;”
Vaccination certificates are intended solely for the purpose of monitoring the effectiveness of the vaccines, as well as their side effects and adverse events.
In addition to this, the other contents are also worth reading and paying attention to.
Portugal – 0.9 % Covid-19 deaths instead of officially 17,000
Thanks to a petition from the Portuguese population, the court in Portugal had to deal with the question of how many people were registered as Covid-19 dead and officially with around 17.000 were estimated to have actually died of Covid-19.
The court took evidence and came up with its Verdict dated May 19, 2021 came to the conclusion that only 152 people died of Covid-19.
According to the usual reading, every deceased person who had a positive PCR test within the last 28 days or was registered as a contact person must be counted as a Covid-19 death.
On November 11th, 2020, the Lisbon Court of Appeal had already dealt with the lawsuit against the quarantine measures that had been ordered based on PCR test results. It highlighted in a 34-page Verdict, based on a number of scientific sources, the quarantine order regarding the questionable validity of the PCR test procedure.
Reporting point for the clarification of deaths after CORONA vaccination
The Union Doctors and Scientists for Health, Freedom and Democracy eV offers a wide range of information, tips and suggestions on its website about the autopsy of deceased people after a corona vaccination. This is also available for download here PDF published. It is explicitly pointed out, among other things, that the autopsy was carried out in accordance with the recommendations of
Prof. Dr. Arne Burkhardt
Reutlingen pathology laboratory
Upper waters 3-7
72764 Reutlingen
are to be carried out.
The association board consists of Prof. Dr. med. Sucharit Bhakdi, specialist in microbiology and infection epidemiology, professor emeritus. the Johannes Gutenberg University Mainz, from 1991 to 2012 head of the local Institute for Medical Microbiology and Hygiene, Dr. med. Ronald Weikl, gynecologist, Prof. Dr. Stefan Homburg, Professor of Public Finance, Leibniz University Hannover and Daniela Folkinger, Psychological Consultant, Teacher, Thurmansbang.
Osteopathy and corona vaccination
Jens Oskamp*, osteopath in Cologne, has written the following patient information, which refers to the risks of vector and mRNA vaccines in connection with osteopathic treatment and therefore causalizes the exclusion of vaccinated people.
„Unfortunately, I have to inform you that I cannot treat people who have received so-called mRNA and vector vaccines against SARS CoV2. In contrast to traditional vaccinations, these are genetic engineering methods that manipulate the body's own cells to produce parts of a virus themselves with the aim of triggering an immune response from the body. These “vaccines” only have emergency approval. There is little or no research into cross-reactions with other medications and therapies. (Video – Prof. Dr. Hockertz, 2020)
The following problems arise in this regard for osteopathic treatments:
It is not yet clear which areas of the body are affected by thrombosis formation. The well-known cerebral vein thrombosis as a side effect occurs because blood flows relatively slowly in this area of the body (Chen et al. 2021). But slow-flowing blood also occurs in other parts of the body. Thrombi can also form there. (Kadkhoda,2021). If, for example, the blood vessels in the venous system of the legs become more permeable through osteopathic techniques, thrombi that have formed can initially become loose, which in the worst case can lead to a pulmonary embolism. Thrombus formation often occurs without symptoms.
Furthermore, it cannot be ruled out that further uncontrolled immune reactions may occur as soon as pathogens are released from the tissue during osteopathic treatment. Normally the immune system can handle it easily. However, an over-reactive immune system can lead to serious complications and destroy the body's own tissue (Vojdania and Kharrazianb, 2020), (Talotta,2021).
Good osteopathic treatment removes blockages in the lymphatic system. As a result of mRNA manipulation, an unnatural amount of specific antibodies is stored there (Room 2020). It cannot be ruled out that significant reactions will occur (Hotez et al. 2020) as soon as these tissues change as part of an osteopathic treatment. The nervous system can also be affected, such as cases of fascial palsy [facial paralysis] (Shemer et al. 2021),(Renould et al, 2021) or eye problems due to a Papilledema (German Ophthalmological Society, 2021) show.
Further problems arise from the nanoparticles used in the mRNA vaccines (Chen et al. 2021). Among other things, they lead to vacuolization (practically edema formation at the cellular level) of certain types of tissue, especially the liver. This is a sign that the corresponding cells have died as a result of reactions with nanoparticles (Video – Dr. Vanessa Schmidt-Krüger, 2021*). Here, too, it is not clear what happens if the fluid from these “edemas” or the dead tissue gets into the bloodstream through osteopathic techniques.
*Local download links to Dr.'s videos Vanessa Schmidt-Krüger are here (Video 1_2) and here (Video 2_2) available.
If you have already undergone genetic modifications using the mRNA/vector vaccines, I ask you to make appointments with me again no earlier than 10 months after these measures. We will then discuss which laboratory tests and imaging procedures are necessary to rule out complications. (e.g. Examination for papilledema - German Ophthalmological Society, 2021)
Multiple injections increase the intensity and likelihood of the relationships described above. This means that I reserve the right to generally refuse treatment even after a period of 10 months.
A strong immune system as an alternative to vaccination requires knowledge!!! I recommend the following video material:
- Dr. rer. nat. Markus Stark – Strengthen the immune system and defenses
- Dr.med.Mathias Rath – End the current pandemic – prevent future pandemics!
- Prof. Dr. Jörg Spitz – Vitamin D – Hype or Hope„
- Jens Oskamp's website (currently being updated)*
Amendment to the Infection Protection and Basic Law
With the one published by the German Bundestag on June 22nd, 2021 Printed matter 19/30938 becomes for the 23.07.2023 the RESTRICTION of the right to physical integrity, which was still guaranteed by the constitution!
' Article 9
Change to the Infection Protection Act
§ 36 paragraph 12 of the Infection Protection Act of July 20, 2000 (BGBl.
I S. 1045), most recently by Article 1 of the Law of May 28, 2021
(BGBl. I p. 1174) has been changed, is worded as follows:
“ (12) One based on paragraph 8 sentence 1 or paragraph 10 sentence 1
The legal regulation issued comes into effect no later than one year after the repeal of the
Determination of the epidemic situation of national concern by the
German Bundestag invalid according to Section 5 Paragraph 1 Sentence 2. Up to hers
A legal regulation issued on the basis of paragraph 8 sentence 1 or paragraph 10 sentence 1 can be changed even after the epidemic situation of national importance has been lifted.
Article 10
Restriction of fundamental rights
By Article 9 become the basic rights of physical integrity (Article 2 paragraph 2 sentence 1 of the Basic Law), freedom of the person
(Article 2 paragraph 2 sentence 2 of the Basic Law), freedom of movement (Article 11
Paragraph 1 of the Basic Law) and the inviolability of the home (Article 13 Paragraph 1 of the Basic Law) restricted.
7. The previous Article 9 becomes Article 11 and paragraph 2 is worded as follows:
(2) “Articles 1, 2, 6, 7 numbers 1, 2 and 4 as well as Article 8 come into force on July 2023.”
Evaluation of 109 studies on wearing masks
On April 20, 2021, an evaluation of 109 studies on the health aspects of wearing masks during pandemic times was carried out International Journal of Environmental Research and Public Health published, the here as PDF in the original (English) and here The German version is available for download.
The researchers came to a result that they themselves did not believe was possible given this level of damage.
In addition to the already known negative effects, the mask-induced exhaustion syndrome (MIES) should be highlighted.
The effects of MIES can include problems with concentration, thinking and speech, a decrease in heart and breathing rates, and depth of breathing, which in turn can cause damage to the blood and coronary arteries and, as a result, neurological and cardiac diseases. Long-term effects are still the subject of ongoing research.
WHO – Change in vaccination recommendations for children
In the version From June 3rd, 2021, the recommendation was not to vaccinate children at the moment, as there was still no reliable evidence to vaccinate children against Covid-19, especially since they, like adolescents, usually have milder cases compared to adults. The usual recommended vaccinations for children should still be continued.
In the current version, published on June 20, 2021, the above passage has been changed to the effect that although the milder courses are pointed out and a vaccination does not necessarily have to be carried out as long as the children are not part of a risk group, more information is still needed in order to do so to make general vaccination recommendations.
Nevertheless, Pfizer BioNTech is defined as suitable for children over 12 years of age. Children between the ages of 12 and 15 as members of risk groups should also be offered this vaccine, along with other priority groups.
As in the old version, it is pointed out that there is not yet enough data from test series with children. As soon as further information becomes available, appropriate recommendations will be issued.
Changed passages are highlighted in yellow in the documents above.
Loud Decision of the STIKO for the 6th update of the COVID-19 vaccination recommendation and the associated scientific justification in Epidemiological Bulletin 23/2021 It is advisable (in line with the current WHO recommendations) to “vaccinate children and adolescents with previous illnesses with the mRNA vaccine Comirnaty (BioNTech/Pfizer) due to an assumed increased risk of a severe course of the COVID-19 disease.” “The use of Comirnaty in children and adolescents aged 12-17 without previous illnesses is currently not generally recommended, but is possible after medical advice and with individual wishes and risk acceptance.”
PEI – Safety Report
The PEI (Paul Ehrlich Institute), a federal agency of the Ministry of Health that is bound by instructions, publishes so-called safety reports at intervals of several weeks regarding the vaccines used and their side effects.
The RKI (Robert Koch Institute), an independent higher federal authority bound by instructions in the sense of Art. 87 Paragraph 3 Sentence 1 GG. It is home to “several scientific commissions, for example the Standing Vaccination Commission, which develops vaccination recommendations. It is also responsible for the content processing and coordination of federal health reporting and for approving the import and use of human embryonic stem cells.”
Summary of cases:
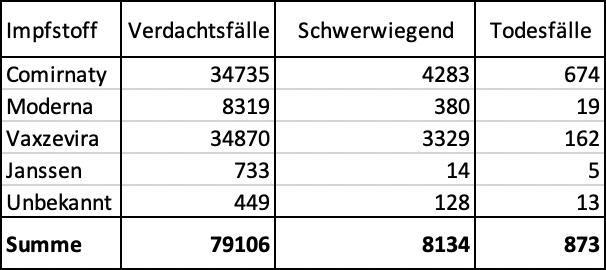
Further information about these authorities can be found on the website Federal Ministry of Health.
The US database VAERS gives the following numbers for the USA:
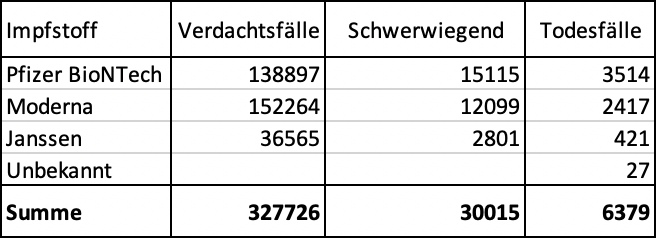
For comparison: since vaccinations against diphtheria, measles, mumps, rubella, polio and tetanus began, 4,050 people have died in the USA. The first diphtheria vaccine was first approved in Germany in 1936, the measles vaccine in the USA in 1963, the mumps and rubella vaccine in 1969, the polio vaccine in 1955, and the tetanus vaccine in 1930.
Ie, on the part of the government as “safe” and “highly effective” Declared Covid-19 vaccines have, based on the data available in the USA, generated 150 % of these deaths within six months (!), which all of the above-mentioned vaccinations together have not achieved in six to eight decades!
Effectiveness of Covid-19 vaccines
As of June 1st, 2021, the RKI announced in its FAQ regarding mRNA vaccines: “How long the vaccination protection lasts is not yet known. Protection does not begin immediately after vaccination, and some vaccinated people remain unprotected.”
Regarding vector-based vaccines, the following is reported: “How long the vaccination protection lasts is not yet known. Protection does not begin immediately after vaccination, and some vaccinated people remain unprotected.”
This raises the question of the extent to which the “adverse side effects” and “deaths” recorded in the PEI safety reports or other databases can be justified if it is openly admitted that neither immediate protection nor protection after multiple vaccinations is given There is no information available about the duration of possible protection.
Archived posts:
FAQ – Corona tests
Post from March 31, 2021 08:43
“Testing” is an almost daily companion these days. And there is often the question of what, which test tests, how and with what significance. Below is a list of the available test procedures and properties:
PCR test
... is used to detect SARS-CoV-2 RNA, i.e. parts of the genetic material of the Covid-19 virus, but not to detect the active, i.e. reproducible, virus.
For detection using a specific fluorescence line, the genetic material contained in the sample must be duplicated. The frequency of the reproduction runs is reflected by the so-called Ct value (cycle threshold value).
Ideally, this Ct value is documented on the laboratory report.
A positive PCR test with a Ct value of 30...35 indicates a low viral load, and a Ct value >35 indicates a very low viral load.
A Ct value of 25, for example, represents a significant viral load. (Source: PCR test – Ct value relevance)
However, since in practice the Ct values are neither documented nor handled in a standardized manner by the different laboratories, and some work with Ct values of 40 or higher (up to 50), PCR test results are neither comparable nor meaningful. The risk of false-positive test results increases as the Ct value increases, with all the negative consequences The Lancet reported in relation to an investigation in the UK.
It is not for nothing that the recommendations are: WHO A PCR test result must always be evaluated in connection with existing disease symptoms and clinical diagnostics.
Antigen test
... is intended to detect an acute infection (protein structures of the corona virus), but requires a high viral load. Therefore, confirmation by a subsequent PCR test (with a low Ct value) is required.
A list of those in Germany with special approval (Expires mid-May 2021) antigen tests placed on the market can be found on the website of the “Federal Institute for Drugs and Medical Devices (BfArM”) BfArM – Antigen tests with special approval.
The test can lead to false positive results if the test equipment has been stored below the recommended storage temperature and is then used.
Antibody test (blood sample – Elisa test / rapid test)
... detect specific antibodies that were produced by the organism in response to the corona virus.
It is irrelevant whether the immune response (formation of the detected antibodies) was due to a previous infection with Covid-19 or a vaccination against Covid-19.
Interview with Dr. Greiner, laboratory diagnostics, Vienna
Covid-19 – What antibodies can say.
Eradicate Covid-19?
Post from March 31, 2021 09:06
Is it possible to eradicate a virus?
– How long did it take until the measles virus was (halfway) “defeated”?
The first measles vaccine with an inactivated split vaccine was approved in the USA in 1963 and has been continuously developed. However, after almost 60 years, measles has still not been eradicated (source: Measles virus).
– How long did it take to eradicate the polio virus?
The first polio vaccination with an inactivated virus was first given in 1955 (source: Polio vaccination), a second one followed in 1960. The WHO declared polio eradicated in 2015, 60 years later(!).
And today it is believed that vaccines developed in a few months, which use vector or mRNA technologies, have not been successfully tested on animals, nor on humans with conventionally designed phase I .. III studies, about their side effects and long-term side effects There are no statements whatsoever, and the products were only placed on the market with emergency approval, which is not stated in the consent forms, that they are capable of defeating or even eradicating a virus - and that within a few months?!
Analogous to the polio/measles viruses, it can be assumed that Covid-19 will take no less than two generations until it is eradicated, or at least under control.
Do you want to continue to impose lockdown, compulsory vaccination (still only indirectly), quarantine, isolation, mask requirement, etc. right up to the digital vaccination certificate?!
The only guaranteed effective way to eradicate the virus remains either a lockdown lasting almost 60 years or removing the host for the virus, i.e. exterminating every person. Then the survival of the virus is eliminated.
Btw: this also explains that a virus always strives not to kill its host in order to be able to continue to multiply. Mutations will therefore always pursue the goal of reproducing in an optimized manner, but not becoming more dangerous for its host. There is no question that viruses can still be fatal for people who are previously ill.
How can one understand the government's back and forth?
Post from 03. April 2021 10:11 p.m
Before attempting to address this question in order to work out one of the possible answers, it is helpful to imagine yourself in 2012.
The German Bundestag commissioned a “risk analysis in civil protection” in 2012, for which the report was published on January 3, 2013 in the form of printed matter 17/12051, which can be found at Risk analysis report – printed matter 17/12051 v. 01/03/2013 can be accessed and downloaded as a PDF.
Under Chapter 2.3 on page 5 you can find the topic “Risk analysis “Pandemic caused by Modi-SARS virus”.
This was followed by a 17-page document entitled “How we get COVID-19 under control” that was classified as “classified information for official use only”, which was still on the website of the Federal Ministry of the Interior on May 20, 2020 How we get COVID-19 under control was available and downloadable. Today it is no longer accessible there, but via this backup link Classified COVID-19 scene paper from the Federal Ministry of the Interior.
All previous, current and future actions by the federal government in connection with COVID-a9 are derived from this.
The government's current efforts to step by step limit all upstream institutions' influence on the Chancellor's decisions, to exclude them, to deprive the legislature of its constitutionally enshrined function, and the structural personnel changes in the Federal Republic of Germany's courts, are all striving in the direction that already started in 1933. This time, however, there are no allies on hand to make it possible to install a new basic law that would put a stop to exactly this endeavor!
EMA – backgrounds
Post from April 20, 2021 02:43
The connections between the function of the EMA (European Medicines Agency) and the career, as well as the resulting conflicts of interest, of EMA President Emer Cooke are presented here, as epochtimes.de reported on April 7th, 2021: EMA President Emer Cooke was a lobbyist for Europe's largest pharmaceutical organization for years
excess mortality
Post from April 20, 2021 08:09
Always happy to address the topic of excess mortality. In addition to the lack of intensive care bed capacity, the argument of excess mortality is a constant companion in justifying the government's measures.
One would think that the Federal Statistical Office's own figures should also be known at federal press conferences (see: Death numbers in March 2021: 11 % below the average of previous years).
Nevertheless, Mr. Hanno Kautz, spokesman for Health Minister Jens Spahn, knows Federal press conference on April 19, 2021 nothing about these numbers. When asked by Mr. Reitschuster: “Mr. Kautz, according to the Federal Statistical Office, we had a low mortality rate last month. In March, 11 percent fewer died than the average for the years 2017 to 2020. How do you explain that?" he simply answers: "I don't comment on numbers that I haven't seen before." and "You quote a very specific number, which I don't know, whose context I don't know. I can’t comment on that at this point.”
The question arises: if these numbers are so uninteresting, and people haven't even noticed them or know them, why are they constantly cited as a justification for increasingly strict measures?